The landscape of human proteins interacting with viruses and other pathogens
- PMID: 18282095
- PMCID: PMC2242834
- DOI: 10.1371/journal.ppat.0040032
The landscape of human proteins interacting with viruses and other pathogens
Abstract
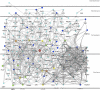
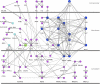
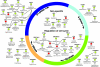
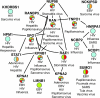
Infectious diseases result in millions of deaths each year. Mechanisms of infection have been studied in detail for many pathogens. However, many questions are relatively unexplored. What are the properties of human proteins that interact with pathogens? Do pathogens interact with certain functional classes of human proteins? Which infection mechanisms and pathways are commonly triggered by multiple pathogens? In this paper, to our knowledge, we provide the first study of the landscape of human proteins interacting with pathogens. We integrate human-pathogen protein-protein interactions (PPIs) for 190 pathogen strains from seven public databases. Nearly all of the 10,477 human-pathogen PPIs are for viral systems (98.3%), with the majority belonging to the human-HIV system (77.9%). We find that both viral and bacterial pathogens tend to interact with hubs (proteins with many interacting partners) and bottlenecks (proteins that are central to many paths in the network) in the human PPI network. We construct separate sets of human proteins interacting with bacterial pathogens, viral pathogens, and those interacting with multiple bacteria and with multiple viruses. Gene Ontology functions enriched in these sets reveal a number of processes, such as cell cycle regulation, nuclear transport, and immune response that participate in interactions with different pathogens. Our results provide the first global view of strategies used by pathogens to subvert human cellular processes and infect human cells. Supplementary data accompanying this paper is available at http://staff.vbi.vt.edu/dyermd/publications/dyer2008a.html.
Conflict of interest statement
Figures


References
-
- LaCount DJ, Vignali M, Chettier R, Phansalkar A, Bell R, et al. A protein interaction network of the malaria parasite Plasmodium falciparum . Nature. 2005;438:103–107. - PubMed
-
- Filippova M, Parkhurst L, Duerksen-Hughes PJ. The human papillomavirus 16 E6 protein binds to Fas-associated death domain and protects cells from Fas-triggered apoptosis. J Biol Chem. 2004;279:25729–25744. - PubMed
-
- Hayashi F, Smith KD, Ozinsky A, Hawn TR, Yi EC, et al. The innate immune response to bacterial flagellin is mediated by toll-like receptor 5. Nature. 2001;410:1099–1103. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

