Double inhibition of XIAP and Bcl-2 axis is beneficial for retrieving sensitivity of renal cell cancer to apoptosis
- PMID: 18283311
- PMCID: PMC2266840
- DOI: 10.1038/sj.bjc.6604268
Double inhibition of XIAP and Bcl-2 axis is beneficial for retrieving sensitivity of renal cell cancer to apoptosis
Abstract
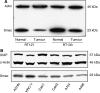
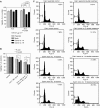
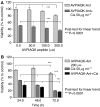
Renal cell carcinoma (RCC) is known to be resistant to chemo- and radiotherapy due to a high apoptotic threshold. Smac and XIAP (X-linked inhibitor of apoptosis protein) proteins were detected in all RCC cell lines and tissue samples examined. We modulated the function of XIAP, either through its constitutional downregulation with an shRNA vector or by applying a Smac-mimicking peptide. Among RCC cell lines, Caki1 expresses the highest levels of XIAP. We transfected Caki1 with XIAP-targeting shRNA vector and generated stable clones. XIAP was knocked down by RNA interference in clone no. 14 by 81.6% and in clone no. 19 by 85.3%. Compared to the parental and mock-transfected cells, neither clone was more sensitive to conventional chemotherapeutic agents, but both clones were more susceptible to Fas stimulation (P<0.0001) and to pharmacological Bcl-2 inhibition (P<0.0001), as well as to a combination of the two (P<0.0001). Mature Smac binds to XIAP via the N-terminal residues, disrupting its interaction with caspases and promoting their activity. We determined that exposure of Caki1 cells to Smac-N7 peptide (AVPIAQK) resulted in a slight but significant decrease in viability (P=0.0031) and potentiated cisplatin's effect (P=0.0027). In contrast with point targeting of XIAP by shRNA, Smac-N7 peptide is active against several IAP (inhibitor of apoptosis protein) family members, which can explain its role in sensitising cells to cisplatin. Our results suggest that multiple targeting of both Bcl-2 and XIAP or, alternatively, of several IAP family members by the Smac-N7 peptide is a potent way to overcome resistance of RCC to apoptosis-triggering treatment modalities, and might be a new tool for molecular targeted therapy.
Figures





Similar articles
-
Effect of the XIAP inhibitor Embelin on TRAIL-induced apoptosis of pancreatic cancer cells.J Surg Res. 2007 Oct;142(2):281-6. doi: 10.1016/j.jss.2007.03.068. Epub 2007 Jul 19. J Surg Res. 2007. PMID: 17640673
-
Novel SMAC-mimetics synergistically stimulate melanoma cell death in combination with TRAIL and Bortezomib.Br J Cancer. 2010 Jun 8;102(12):1707-16. doi: 10.1038/sj.bjc.6605687. Epub 2010 May 11. Br J Cancer. 2010. PMID: 20461078 Free PMC article.
-
Disturbed balance of expression between XIAP and Smac/DIABLO during tumour progression in renal cell carcinomas.Br J Cancer. 2004 Oct 4;91(7):1349-57. doi: 10.1038/sj.bjc.6602127. Br J Cancer. 2004. PMID: 15328523 Free PMC article.
-
Targeting XIAP for Promoting Cancer Cell Death-The Story of ARTS and SMAC.Cells. 2020 Mar 9;9(3):663. doi: 10.3390/cells9030663. Cells. 2020. PMID: 32182843 Free PMC article. Review.
-
Targeting Inhibitor of Apoptosis Proteins to Overcome Chemotherapy Resistance-A Marriage between Targeted Therapy and Cytotoxic Chemotherapy.Int J Mol Sci. 2023 Aug 29;24(17):13385. doi: 10.3390/ijms241713385. Int J Mol Sci. 2023. PMID: 37686191 Free PMC article. Review.
Cited by
-
Potential biological process of X-linked inhibitor of apoptosis protein in renal cell carcinoma based upon differential protein expression analysis.Oncol Lett. 2018 Jan;15(1):821-832. doi: 10.3892/ol.2017.7383. Epub 2017 Nov 9. Oncol Lett. 2018. PMID: 29403558 Free PMC article.
-
Uncoupling of PUMA Expression and Apoptosis Contributes to Functional Heterogeneity in Renal Cell Carcinoma - Prognostic and Translational Implications.Transl Oncol. 2015 Dec;8(6):480-6. doi: 10.1016/j.tranon.2015.11.003. Transl Oncol. 2015. PMID: 26692529 Free PMC article.
-
XIAP underlies apoptosis resistance of renal cell carcinoma cells.Mol Med Rep. 2018 Jan;17(1):125-130. doi: 10.3892/mmr.2017.7925. Epub 2017 Oct 27. Mol Med Rep. 2018. PMID: 29115633 Free PMC article.
-
Down-regulation of cIAP2 enhances 5-FU sensitivity through the apoptotic pathway in human colon cancer cells.Cancer Sci. 2009 May;100(5):903-13. doi: 10.1111/j.1349-7006.2009.01112.x. Epub 2009 Mar 2. Cancer Sci. 2009. PMID: 19302291 Free PMC article.
-
Isobaric tags for relative and absolute quantitation-based quantitative proteomic analysis of X-linked inhibitor of apoptosis and H2AX in etoposide-induced renal cell carcinoma apoptosis.Chin Med J (Engl). 2019 Dec 20;132(24):2941-2949. doi: 10.1097/CM9.0000000000000553. Chin Med J (Engl). 2019. PMID: 31855962 Free PMC article.
References
-
- Arnt CR, Chiorean MV, Heldebrant MP, Gores GJ, Kaufmann SH (2002) Synthetic Smac/DIABLO peptides enhance the effects of chemotherapeutic agents by binding XIAP and cIAP1 in situ. J Biol Chem 277: 44236–44243 - PubMed
-
- Bilim V, Kasahara T, Hara N, Takahashi K, Tomita Y (2003) Role of XIAP in the malignant phenotype of transitional cell cancer (TCC) and therapeutic activity of XIAP antisense oligonucleotides against multidrug-resistant TCC in vitro. Int J Cancer 103: 29–37 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

