A novel function for hydroxyproline oxidase in apoptosis through generation of reactive oxygen species
- PMID: 18287100
- PMCID: PMC2447650
- DOI: 10.1074/jbc.M702181200
A novel function for hydroxyproline oxidase in apoptosis through generation of reactive oxygen species
Abstract
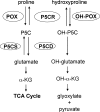
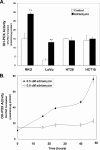
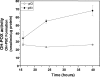
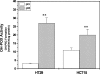
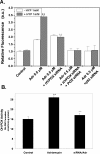
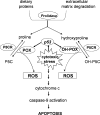
Proline and hydroxyproline are metabolized by distinct pathways. Proline is important for protein synthesis, as a source of glutamate, arginine, and tricarboxylic acid cycle intermediates, and for participating in a metabolic cycle that shuttles redox equivalents between mitochondria and cytosol. Hydroxyproline, in contrast, is not reutilized for protein synthesis. The first steps in the degradation of proline and hydroxyproline are catalyzed by proline oxidase (POX) and hydroxyproline oxidase (OH-POX), respectively. Because it is well documented that POX is induced by p53 and plays a role in apoptosis, we considered whether OH-POX also participates in the response to cytotoxic stress. In LoVo and RKO cells, which respond to adriamycin with a p53-mediated induction of POX and generation of reactive oxygen species, we found that adriamycin also induced OH-POX gene expression and markedly increased OH-POX catalytic activity, and this increase in activity was not observed in the cell lines HT29 and HCT15, which do not have a functional p53. We also observed an increase in reactive oxygen species generation and activation of caspase-9 with adriamycin in a hydroxyproline-dependent manner. Therefore, we hypothesize that OH-POX plays a role analogous to POX in growth regulation, ROS generation, and activation of the apoptotic cascade.
Figures


References
-
- Pandhare, J., Cooper, S. K., and Phang, J. M. (2006) J. Biol. Chem. 281 2044–2052 - PubMed
-
- Maxwell, S. A., and Rivera, A. (2003) J. Biol. Chem. 278 9784–9789 - PubMed
-
- Donald, S. P., Sun, X. Y., Hu, C. A., Yu, J., Mei, J. M., Valle, D., and Phang, J. M. (2001) Cancer Res. 61 1810–1815 - PubMed
-
- Liu, Y., Borchert, G. L., Surazynski, A., Hu, C. A., and Phang, J. M. (2006) Oncogene 25 5640–5647 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

