FBXO25-associated nuclear domains: a novel subnuclear structure
- PMID: 18287534
- PMCID: PMC2366848
- DOI: 10.1091/mbc.e07-08-0815
FBXO25-associated nuclear domains: a novel subnuclear structure
Abstract
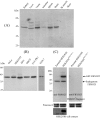
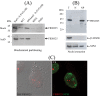
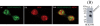
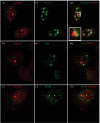
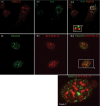
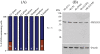
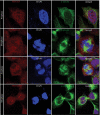
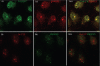
Skp1, Cul1, Rbx1, and the FBXO25 protein form a functional ubiquitin ligase complex. Here, we investigate the cellular distribution of FBXO25 and its colocalization with some nuclear proteins by using immunochemical and biochemical approaches. FBXO25 was monitored with affinity-purified antibodies raised against the recombinant fragment spanning residues 2-62 of the FBXO25 sequence. FBXO25 protein was expressed in all mouse tissues tested except striated muscle, as indicated by immunoblot analysis. Confocal analysis revealed that the endogenous FBXO25 was partially concentrated in a novel dot-like nuclear domain that is distinct from clastosomes and other well-characterized structures. These nuclear compartments contain a high concentration of ubiquitin conjugates and at least two other components of the ubiquitin-proteasome system: 20S proteasome and Skp1. We propose to name these compartments FBXO25-associated nuclear domains. Interestingly, inhibition of transcription by actinomycin D or heat-shock treatment drastically affected the nuclear organization of FBXO25-containing structures, indicating that they are dynamic compartments influenced by the transcriptional activity of the cell. Also, we present evidences that an FBXO25-dependent ubiquitin ligase activity prevents aggregation of recombinant polyglutamine-containing huntingtin protein in the nucleus of human embryonic kidney 293 cells, suggesting that this protein can be a target for the nuclear FBXO25 mediated ubiquitination.
Figures




References
-
- Ang X. L., Harper J. W. Interwoven ubiquitination oscillators and control of cell cycle transitions. Oncogene. 2005;17:2860–2870. - PubMed
-
- Ardley H. C., Robinson P. A. E3 ubiquitin ligases. Essays Biochem. 2005;41:15–30. - PubMed
-
- Ausubel F. M., Brent R., Kingston R. E., Moore D. D, Seidman J. G., Smith J. A., Struhl K. Short Protocols in Molecular Biology. 3rd ed. New York: John Wiley & Sons; 1997.
-
- Carmo-Fonseca M., Mendes-Soares L., Campos I. To be or not to be in the nucleolus. Nat. Cell Biol. 2000;2:E107–E112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

