Integrin affinity modulation in angiogenesis
- PMID: 18287811
- PMCID: PMC2677212
- DOI: 10.4161/cc.7.3.5234
Integrin affinity modulation in angiogenesis
Abstract
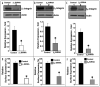
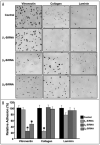
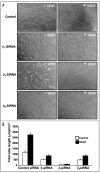
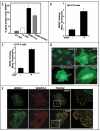
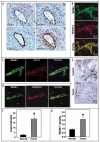
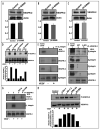
Integrins, transmembrane glycoprotein receptors, play vital roles in pathological angiogenesis, but their precise regulatory functions are not completely understood and remain controversial. This study aims to assess the regulatory functions of individual beta subunits of endothelial integrins in angiogenic responses induced by vascular endothelial growth factor (VEGF). Inhibition of expression of beta(1), beta(3), or beta(5) integrins in endothelial cells resulted in down regulation of EC adhesion and migration on the primary ligand for the corresponding integrin receptor, while no effects on the recognition of other ligands were detected. Although inhibition of expression of each subunit substantially affected capillary growth stimulated by VEGF, the loss of beta(3) integrin was the most inhibitory.
Figures

Similar articles
-
The alpha(1)beta(1) and alpha(2)beta(1) integrins provide critical support for vascular endothelial growth factor signaling, endothelial cell migration, and tumor angiogenesis.Am J Pathol. 2002 Jan;160(1):195-204. doi: 10.1016/s0002-9440(10)64363-5. Am J Pathol. 2002. PMID: 11786413 Free PMC article.
-
Integrin regulation by vascular endothelial growth factor in human brain microvascular endothelial cells: role of alpha6beta1 integrin in angiogenesis.J Biol Chem. 2006 Dec 29;281(52):40450-60. doi: 10.1074/jbc.M607525200. Epub 2006 Nov 2. J Biol Chem. 2006. PMID: 17085437
-
Elevated Flk1 (vascular endothelial growth factor receptor 2) signaling mediates enhanced angiogenesis in beta3-integrin-deficient mice.Cancer Res. 2004 Dec 1;64(23):8643-50. doi: 10.1158/0008-5472.CAN-04-2760. Cancer Res. 2004. PMID: 15574772
-
Endothelial cell integrins and COX-2: mediators and therapeutic targets of tumor angiogenesis.Biochim Biophys Acta. 2004 Mar 4;1654(1):51-67. doi: 10.1016/j.bbcan.2003.09.003. Biochim Biophys Acta. 2004. PMID: 14984767 Review.
-
Role of alpha v integrins during angiogenesis.Cancer J. 2000 May;6 Suppl 3:S245-9. Cancer J. 2000. PMID: 10874494 Review.
Cited by
-
Integrin β3 crosstalk with VEGFR accommodating tyrosine phosphorylation as a regulatory switch.PLoS One. 2012;7(2):e31071. doi: 10.1371/journal.pone.0031071. Epub 2012 Feb 17. PLoS One. 2012. PMID: 22363548 Free PMC article.
-
c-Abl mediates endothelial apoptosis induced by inhibition of integrins alphavbeta3 and alphavbeta5 and by disruption of actin.Blood. 2010 Apr 1;115(13):2709-18. doi: 10.1182/blood-2009-05-223776. Epub 2010 Feb 2. Blood. 2010. PMID: 20124512 Free PMC article.
-
Integrin-mediated cell-matrix interaction in physiological and pathological blood vessel formation.J Oncol. 2012;2012:125278. doi: 10.1155/2012/125278. Epub 2011 Sep 18. J Oncol. 2012. PMID: 21941547 Free PMC article.
-
Beyond classical collagen: basement membrane collagen IV in age-associated lung diseases.Eur Respir Rev. 2025 Jul 23;34(177):240192. doi: 10.1183/16000617.0192-2024. Print 2025 Jul. Eur Respir Rev. 2025. PMID: 40701642 Free PMC article. Review.
-
Biological production of an integrin αvβ3 targeting imaging probe and functional verification.Biomed Res Int. 2015;2015:681012. doi: 10.1155/2015/681012. Epub 2015 Jan 15. Biomed Res Int. 2015. PMID: 25654118 Free PMC article.
References
-
- Plow EF, Haas TA, Zhang L, Loftus J, Smith JW. Ligand binding to integrins. J Biol Chem. 2000;275:21785–8. - PubMed
-
- Arnaout MA, Goodman SL, Xiong JP. Coming to grips with integrin binding to ligands. Curr Opin Cell Biol. 2002;14:641–51. - PubMed
-
- Echarri A, Del Pozo MA. Caveolae internalization regulates integrin-dependent signaling pathways. Cell Cycle. 2006;5:2179–82. - PubMed
-
- Fang K, Fu W, Beardsley AR, Sun X, Lisanti MP, Liu J. Overexpression of caveolin-1 inhibits endothelial cell proliferation by arresting the cell cycle at G0/G1 phase. Cell Cycle. 2007;6:199–204. - PubMed
-
- Ruoslahti E. Integrin signaling and matrix assembly. Tumour Biol. 1996;17:117–24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
