MYC in mammalian epidermis: how can an oncogene stimulate differentiation?
- PMID: 18292777
- PMCID: PMC2494614
- DOI: 10.1038/nrc2328
MYC in mammalian epidermis: how can an oncogene stimulate differentiation?
Erratum in
- Nat Rev Cancer. 2008 Apr;8(4):316
Abstract
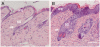
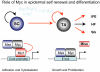
MYC in human epidermal stem cells can stimulate differentiation rather than uncontrolled proliferation. This discovery was, understandably, greeted with scepticism by researchers. However, subsequent studies have confirmed that MYC can stimulate epidermal stem cells to differentiate and have shed light on the underlying mechanisms. Two concepts that are relevant to cancer have emerged: first, MYC regulates similar genes in different cell types, but the biological consequences are context-dependent; and second, MYC activation is not a simple 'on/off' switch - the cellular response depends on the strength and duration of MYC activity, which in turn is affected by the many cofactors and regulatory pathways with which MYC interacts.
Figures



Similar articles
-
c-Myc promotes differentiation of human epidermal stem cells.Genes Dev. 1997 Nov 1;11(21):2869-82. doi: 10.1101/gad.11.21.2869. Genes Dev. 1997. PMID: 9353256 Free PMC article.
-
Reversible activation of c-Myc in skin: induction of a complex neoplastic phenotype by a single oncogenic lesion.Mol Cell. 1999 May;3(5):565-77. doi: 10.1016/s1097-2765(00)80350-0. Mol Cell. 1999. PMID: 10360173
-
Lack of cyclin-dependent kinase 4 inhibits c-myc tumorigenic activities in epithelial tissues.Mol Cell Biol. 2004 Sep;24(17):7538-47. doi: 10.1128/MCB.24.17.7538-7547.2004. Mol Cell Biol. 2004. PMID: 15314163 Free PMC article.
-
Gene Transactivation and Transrepression in MYC-Driven Cancers.Int J Mol Sci. 2021 Mar 27;22(7):3458. doi: 10.3390/ijms22073458. Int J Mol Sci. 2021. PMID: 33801599 Free PMC article. Review.
-
c-Myc and activated Ras during skin tumorigenesis: cooperation at the cancer stem cell level?Ernst Schering Found Symp Proc. 2006;(5):13-26. doi: 10.1007/2789_2007_042. Ernst Schering Found Symp Proc. 2006. PMID: 17939293 Review.
Cited by
-
Deciphering the role of nuclear and cytoplasmic IKKα in skin cancer.Oncotarget. 2016 May 17;7(20):29531-47. doi: 10.18632/oncotarget.8792. Oncotarget. 2016. PMID: 27121058 Free PMC article.
-
Normal and Neoplastic Growth Suppression by the Extended Myc Network.Cells. 2022 Feb 21;11(4):747. doi: 10.3390/cells11040747. Cells. 2022. PMID: 35203395 Free PMC article. Review.
-
p53 Loss in Breast Cancer Leads to Myc Activation, Increased Cell Plasticity, and Expression of a Mitotic Signature with Prognostic Value.Cell Rep. 2019 Jan 15;26(3):624-638.e8. doi: 10.1016/j.celrep.2018.12.071. Cell Rep. 2019. PMID: 30650356 Free PMC article.
-
Concise Review: Control of Cell Fate Through Cell Cycle and Pluripotency Networks.Stem Cells. 2016 Jun;34(6):1427-36. doi: 10.1002/stem.2345. Epub 2016 Mar 16. Stem Cells. 2016. PMID: 26889666 Free PMC article. Review.
-
Multi-scale Modelling for Threshold Dependent Differentiation.Math Model Nat Phenom. 2009 Jun 1;4(4):103-117. doi: 10.1051/mmnp/20094403. Math Model Nat Phenom. 2009. PMID: 20622931 Free PMC article.
References
-
- Owens DM, Watt FM. Contribution of stem cells and differentiated cells to epidermal tumours. Nat. Rev. Cancer. 2003;3:444–451. - PubMed
-
- Watt FM, Lo Celso C, Silva-Vargas V. Epidermal stem cells: an update. Curr. Opin. Genet. Dev. 2006;16:518–524. - PubMed
-
- Jones PH, Simons BD, Watt FM. Sic transit Gloria. Farewell to the epidermal transit amplifying cell? Cell Stem Cell. 2007;1:371–381. - PubMed
-
- Boukamp P. Non-melanoma skin cancer: what drives tumor development and progression? Carcinogenesis. 2005;26:1657–1667. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

