PIKfyve: Partners, significance, debates and paradoxes
- PMID: 18304842
- PMCID: PMC2491398
- DOI: 10.1016/j.cellbi.2008.01.006
PIKfyve: Partners, significance, debates and paradoxes
Abstract
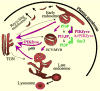
Key components of membrane trafficking and signaling machinery in eukaryotic cells are proteins that bind or synthesize phosphoinositides. PIKfyve, a product of an evolutionarily conserved single-copy gene has both these features. It binds to membrane phosphatidylinositol (PtdIns)3P and synthesizes PtdIns(3,5)P2 and PtdIns5P. Molecular functions of PIKfyve are elusive but recent advances are consistent with a key role in the course of endosomal transport. PIKfyve dysfunction induces endosome enlargement and profound cytoplasmic vacuolation, likely as a result of impaired normal endosome processing and membrane exit out of endosomes. Multicellular organisms with genetically impaired function of PIKfyve or that of the PIKfyve protein partners regulating PtdIns(3,5)P2 homeostasis display severe disorders, including embryonic/perinatal death. This review describes recent advances on PIKfyve functionality in higher eukaryotes, with particular reference to biochemical and genetic insights in PIKfyve protein partners.
Figures

Similar articles
-
Core protein machinery for mammalian phosphatidylinositol 3,5-bisphosphate synthesis and turnover that regulates the progression of endosomal transport. Novel Sac phosphatase joins the ArPIKfyve-PIKfyve complex.J Biol Chem. 2007 Aug 17;282(33):23878-91. doi: 10.1074/jbc.M611678200. Epub 2007 Jun 7. J Biol Chem. 2007. PMID: 17556371
-
Active vacuolar H+ ATPase and functional cycle of Rab5 are required for the vacuolation defect triggered by PtdIns(3,5)P2 loss under PIKfyve or Vps34 deficiency.Am J Physiol Cell Physiol. 2016 Sep 1;311(3):C366-77. doi: 10.1152/ajpcell.00104.2016. Epub 2016 Jun 22. Am J Physiol Cell Physiol. 2016. PMID: 27335171 Free PMC article.
-
Apilimod, a candidate anticancer therapeutic, arrests not only PtdIns(3,5)P2 but also PtdIns5P synthesis by PIKfyve and induces bafilomycin A1-reversible aberrant endomembrane dilation.PLoS One. 2018 Sep 21;13(9):e0204532. doi: 10.1371/journal.pone.0204532. eCollection 2018. PLoS One. 2018. PMID: 30240452 Free PMC article.
-
PIKfyve and its Lipid products in health and in sickness.Curr Top Microbiol Immunol. 2012;362:127-62. doi: 10.1007/978-94-007-5025-8_7. Curr Top Microbiol Immunol. 2012. PMID: 23086417 Review.
-
PtdIns5P: news and views of its appearance, disappearance and deeds.Arch Biochem Biophys. 2013 Oct 15;538(2):171-80. doi: 10.1016/j.abb.2013.07.023. Epub 2013 Aug 2. Arch Biochem Biophys. 2013. PMID: 23916588 Free PMC article. Review.
Cited by
-
The phosphatidylinositol-3-phosphate 5-kinase inhibitor apilimod blocks filoviral entry and infection.PLoS Negl Trop Dis. 2017 Apr 12;11(4):e0005540. doi: 10.1371/journal.pntd.0005540. eCollection 2017 Apr. PLoS Negl Trop Dis. 2017. PMID: 28403145 Free PMC article.
-
ArPIKfyve homomeric and heteromeric interactions scaffold PIKfyve and Sac3 in a complex to promote PIKfyve activity and functionality.J Mol Biol. 2008 Dec 26;384(4):766-79. doi: 10.1016/j.jmb.2008.10.009. Epub 2008 Oct 11. J Mol Biol. 2008. PMID: 18950639 Free PMC article.
-
The PIKfyve inhibitor YM201636 blocks the continuous recycling of the tight junction proteins claudin-1 and claudin-2 in MDCK cells.PLoS One. 2012;7(3):e28659. doi: 10.1371/journal.pone.0028659. Epub 2012 Mar 1. PLoS One. 2012. PMID: 22396724 Free PMC article.
-
Endosomal maturation, Rab7 GTPase and phosphoinositides in African swine fever virus entry.PLoS One. 2012;7(11):e48853. doi: 10.1371/journal.pone.0048853. Epub 2012 Nov 1. PLoS One. 2012. PMID: 23133661 Free PMC article.
-
ArPIKfyve regulates Sac3 protein abundance and turnover: disruption of the mechanism by Sac3I41T mutation causing Charcot-Marie-Tooth 4J disorder.J Biol Chem. 2010 Aug 27;285(35):26760-26764. doi: 10.1074/jbc.C110.154658. Epub 2010 Jul 14. J Biol Chem. 2010. PMID: 20630877 Free PMC article.
References
-
- Babst M. A protein’s final ESCRT. Traffic. 2005;6:2–9. - PubMed
-
- Balla T. Phosphoinositide-derived messengers in endocrine signaling. J Endocrinol. 2006;188:135–53. - PubMed
-
- Banfi H, Downes CP, Rittenhouse SE. Biphasic activation of PKBα/Akt in platelets. Evidence for stimulation both by phosphatidylinositol 3,4-bisphosphate, produced via a novel pathway, and by phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem. 1998;273:11630–7. - PubMed
-
- Berger P, Bonneick S, Willi S, Wymann M, Suter U. Loss of phosphatase activity in myotubularin-related protein 2 is associated with Charcot-Marie-Tooth disease type 4B1. Human Mol Genetics. 2002;11:11569–79. - PubMed
-
- Berwick DC, Dell GC, Welsh GI, Heesom KJ, Hers I, Fletcher LM, et al. Protein kinase B phosphorylation of PIKfyve regulates the trafficking of GLUT4 vesicles. J Cell Sci. 2004;117:5985–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

