Identification of AglE, a second glycosyltransferase involved in N glycosylation of the Haloferax volcanii S-layer glycoprotein
- PMID: 18310347
- PMCID: PMC2347396
- DOI: 10.1128/JB.00056-08
Identification of AglE, a second glycosyltransferase involved in N glycosylation of the Haloferax volcanii S-layer glycoprotein
Abstract
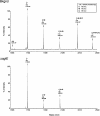
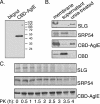
Archaea, like Eukarya and Bacteria, are able to N glycosylate select protein targets. However, in contrast to relatively advanced understanding of the eukaryal N glycosylation process and the information being amassed on the bacterial process, little is known of this posttranslational modification in Archaea. Toward remedying this situation, the present report continues ongoing efforts to identify components involved in the N glycosylation of the Haloferax volcanii S-layer glycoprotein. By combining gene deletion together with mass spectrometry, AglE, originally identified as a homologue of murine Dpm1, was shown to play a role in the addition of the 190-Da sugar subunit of the novel pentasaccharide decorating the S-layer glycoprotein. Topological analysis of an AglE-based chimeric reporter assigns AglE as an integral membrane protein, with its N terminus and putative active site facing the cytoplasm. These finding, therefore, contribute to the developing picture of the N glycosylation pathway in Archaea.
Figures


References
-
- Abu-Qarn, M., and J. Eichler. 2006. Protein N glycosylation in Archaea: defining Haloferax volcanii genes involved in S-layer glycoprotein glycosylation. Mol. Microbiol. 61511-525. - PubMed
-
- Abu-Qarn, M., S. Yurist, A. Giordano, A. Trauner, H. R. Morris, P. Hitchen, O. Medalia, A. Dell, and J. Eichler. 2007. Haloferax volcanii AglB and AglD are involved in N glycosylation of the S-layer glycoprotein and proper assembly of the surface layer. J. Mol. Biol. 3741224-1236. - PubMed
-
- Ashida, H., Y. Maeda, and T. Kinoshita. 2006. DPM1, the catalytic subunit of dolichol-phosphate mannose synthase, is tethered to and stabilized on the endoplasmic reticulum membrane by DPM3. J. Biol. Chem. 281896-904. - PubMed
-
- Baliga, N. S., R. Bonneau, M. T. Tacciotti, M. Pan, G. Glusman, E. W. Deutsch, P. Shannon, Y. Chui, R. S. Weng, R. R. Gan, P. Hung, S. V. Date, E. Marcotte, L. Hood, and W. V. Ng. 2004. Genome sequence of Haloarcula marismortui: a halophilic archaeon from the Dead Sea. Genome Res. 142221-2234. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

