Aberrant mucin assembly in mice causes endoplasmic reticulum stress and spontaneous inflammation resembling ulcerative colitis
- PMID: 18318598
- PMCID: PMC2270292
- DOI: 10.1371/journal.pmed.0050054
Aberrant mucin assembly in mice causes endoplasmic reticulum stress and spontaneous inflammation resembling ulcerative colitis
Abstract
Background: MUC2 mucin produced by intestinal goblet cells is the major component of the intestinal mucus barrier. The inflammatory bowel disease ulcerative colitis is characterized by depleted goblet cells and a reduced mucus layer, but the aetiology remains obscure. In this study we used random mutagenesis to produce two murine models of inflammatory bowel disease, characterised the basis and nature of the inflammation in these mice, and compared the pathology with human ulcerative colitis.
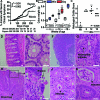
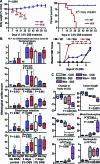
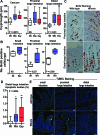
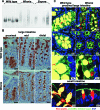
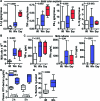
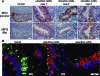
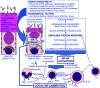
Methods and findings: By murine N-ethyl-N-nitrosourea mutagenesis we identified two distinct noncomplementing missense mutations in Muc2 causing an ulcerative colitis-like phenotype. 100% of mice of both strains developed mild spontaneous distal intestinal inflammation by 6 wk (histological colitis scores versus wild-type mice, p < 0.01) and chronic diarrhoea. Monitoring over 300 mice of each strain demonstrated that 25% and 40% of each strain, respectively, developed severe clinical signs of colitis by age 1 y. Mutant mice showed aberrant Muc2 biosynthesis, less stored mucin in goblet cells, a diminished mucus barrier, and increased susceptibility to colitis induced by a luminal toxin. Enhanced local production of IL-1beta, TNF-alpha, and IFN-gamma was seen in the distal colon, and intestinal permeability increased 2-fold. The number of leukocytes within mesenteric lymph nodes increased 5-fold and leukocytes cultured in vitro produced more Th1 and Th2 cytokines (IFN-gamma, TNF-alpha, and IL-13). This pathology was accompanied by accumulation of the Muc2 precursor and ultrastructural and biochemical evidence of endoplasmic reticulum (ER) stress in goblet cells, activation of the unfolded protein response, and altered intestinal expression of genes involved in ER stress, inflammation, apoptosis, and wound repair. Expression of mutated Muc2 oligomerisation domains in vitro demonstrated that aberrant Muc2 oligomerisation underlies the ER stress. In human ulcerative colitis we demonstrate similar accumulation of nonglycosylated MUC2 precursor in goblet cells together with ultrastructural and biochemical evidence of ER stress even in noninflamed intestinal tissue. Although our study demonstrates that mucin misfolding and ER stress initiate colitis in mice, it does not ascertain the genetic or environmental drivers of ER stress in human colitis.
Conclusions: Characterisation of the mouse models we created and comparison with human disease suggest that ER stress-related mucin depletion could be a fundamental component of the pathogenesis of human colitis and that clinical studies combining genetics, ER stress-related pathology and relevant environmental epidemiology are warranted.
Conflict of interest statement
Figures






References
-
- Herrmann A, Davies JR, Lindell G, Martensson S, Packer NH, et al. Studies on the “insoluble” glycoprotein complex from human colon. Identification of reduction-insensitive MUC2 oligomers and C-terminal cleavage. J Biol Chem. 1999;274:15828–15836. - PubMed
-
- Godl K, Johansson ME, Lidell ME, Morgelin M, Karlsson H, et al. The N terminus of the MUC2 mucin forms trimers that are held together within a trypsin-resistant core fragment. J Biol Chem. 2002;277:47248–47256. - PubMed
-
- Asker N, Axelsson MAB, Olofsson SO, Hansson GC. Dimerization of the human MUC2 mucin in the endoplasmic reticulum is followed by a N-glycosylation-dependent transfer of the mono- and dimers to the Golgi apparatus. J Biol Chem. 1998;273:18857–18863. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

