Proteolysis of AKAP121 regulates mitochondrial activity during cellular hypoxia and brain ischaemia
- PMID: 18323779
- PMCID: PMC2323260
- DOI: 10.1038/emboj.2008.33
Proteolysis of AKAP121 regulates mitochondrial activity during cellular hypoxia and brain ischaemia
Abstract
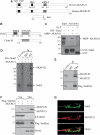
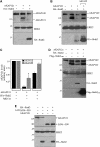
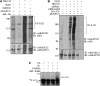
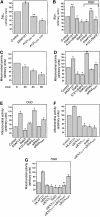
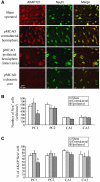
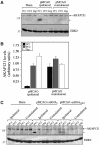
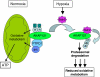
A-kinase anchor protein 121 (AKAP121) assembles a multivalent signalling complex on the outer mitochondrial membrane that controls persistence and amplitude of cAMP and src signalling to mitochondria, and plays an essential role in oxidative metabolism and cell survival. Here, we show that AKAP121 levels are regulated post-translationally by the ubiquitin/proteasome pathway. Seven In-Absentia Homolog 2 (Siah2), an E3-ubiquitin ligase whose expression is induced in hypoxic conditions, formed a complex and degraded AKAP121. In addition, we show that overexpression of Siah2 or oxygen and glucose deprivation (OGD) promotes Siah2-mediated ubiquitination and proteolysis of AKAP121. Upregulation of Siah2, by modulation of the cellular levels of AKAP121, significantly affects mitochondrial activity assessed as mitochondrial membrane potential and oxidative capacity. Also during cerebral ischaemia, AKAP121 is degraded in a Siah2-dependent manner. These findings reveal a novel mechanism of attenuation of cAMP/PKA signaling, which occurs at the distal sites of signal generation mediated by proteolysis of an AKAP scaffold protein. By regulating the stability of AKAP121-signalling complex at mitochondria, cells efficiently and rapidly adapt oxidative metabolism to fluctuations in oxygen availability.
Figures


References
-
- Affaitati A, Cardone L, Carlucci A, de Cristofaro T, Ginsberg MD, Varrone S, Gottesman ME, Avvedimento VE, Feliciello A (2003) Essential role of A-kinase anchor protein 121 for cAMP signalling to mitochondria. J Biol Chem 278: 4286–4294 - PubMed
-
- Cardone L, Carlucci A, Affaitati A, Livigni A, DeCristofaro T, Garbi C, Varrone S, Ullrich A, Gottesman ME, Avvedimento EV, Feliciello A (2004) Mitochondrial AKAP121 binds and targets protein tyrosine phosphatase D1, a novel positive regulator of src signalling. Mol Cell Biol 24: 4613–4626 - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

