The lysophospholipid acyltransferase antagonist CI-976 inhibits a late step in COPII vesicle budding
- PMID: 18331383
- PMCID: PMC2432463
- DOI: 10.1111/j.1600-0854.2008.00711.x
The lysophospholipid acyltransferase antagonist CI-976 inhibits a late step in COPII vesicle budding
Abstract
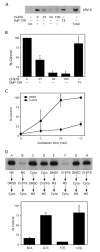
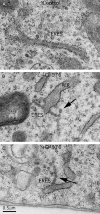
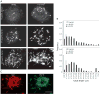
The mechanism of coat protein (COP)II vesicle fission from the endoplasmic reticulum (ER) remains unclear. Lysophospholipid acyltransferases (LPATs) catalyze the conversion of various lysophospholipids to phospholipids, a process that can promote spontaneous changes in membrane curvature. Here, we show that 2,2-methyl-N-(2,4,6,-trimethoxyphenyl)dodecanamide (CI-976), a potent LPAT inhibitor, reversibly inhibited export from the ER in vivo and the formation of COPII vesicles in vitro. Moreover, CI-976 caused the rapid and reversible accumulation of cargo at ER exit sites (ERESs) containing the COPII coat components Sec23/24 and Sec13/31 and a marked enhancement of Sar1p-mediated tubule formation from ERESs, suggesting that CI-976 inhibits the fission of assembled COPII budding elements. These results identify a small molecule inhibitor of a very late step in COPII vesicle formation, consistent with fission inhibition, and demonstrate that this step is likely facilitated by an ER-associated LPAT.
Figures
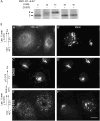
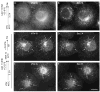
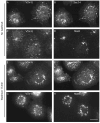
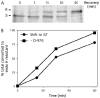
Similar articles
-
Use of acyltransferase inhibitors to block vesicular traffic between the ER and Golgi complex.Methods Enzymol. 2005;404:115-25. doi: 10.1016/S0076-6879(05)04012-7. Methods Enzymol. 2005. PMID: 16413263
-
Sar1p N-terminal helix initiates membrane curvature and completes the fission of a COPII vesicle.Cell. 2005 Aug 26;122(4):605-17. doi: 10.1016/j.cell.2005.07.025. Cell. 2005. PMID: 16122427
-
Activation of phospholipase D by the small GTPase Sar1p is required to support COPII assembly and ER export.EMBO J. 2003 Aug 15;22(16):4059-69. doi: 10.1093/emboj/cdg390. EMBO J. 2003. PMID: 12912905 Free PMC article.
-
A structural view of the COPII vesicle coat.Curr Opin Struct Biol. 2004 Apr;14(2):147-53. doi: 10.1016/j.sbi.2004.02.002. Curr Opin Struct Biol. 2004. PMID: 15093828 Review.
-
Mechanisms of COPII vesicle formation and protein sorting.FEBS Lett. 2007 May 22;581(11):2076-82. doi: 10.1016/j.febslet.2007.01.091. Epub 2007 Feb 14. FEBS Lett. 2007. PMID: 17316621 Review.
Cited by
-
A novel lysophosphatidylcholine acyl transferase activity is expressed by peroxiredoxin 6.J Lipid Res. 2016 Apr;57(4):587-96. doi: 10.1194/jlr.M064758. Epub 2016 Feb 1. J Lipid Res. 2016. PMID: 26830860 Free PMC article.
-
Regulation of the Golgi complex by phospholipid remodeling enzymes.Biochim Biophys Acta. 2012 Aug;1821(8):1078-88. doi: 10.1016/j.bbalip.2012.04.004. Epub 2012 Apr 22. Biochim Biophys Acta. 2012. PMID: 22562055 Free PMC article. Review.
-
Structural and functional analysis of the globular head domain of p115 provides insight into membrane tethering.J Mol Biol. 2009 Aug 7;391(1):26-41. doi: 10.1016/j.jmb.2009.04.062. Epub 2009 May 4. J Mol Biol. 2009. PMID: 19414022 Free PMC article.
-
2-Polyunsaturated acyl lysophosphatidylethanolamine attenuates inflammatory response in zymosan A-induced peritonitis in mice.Lipids. 2011 Oct;46(10):893-906. doi: 10.1007/s11745-011-3589-2. Epub 2011 Jul 10. Lipids. 2011. PMID: 21744277
-
Chemical Manipulation of the Endosome Trafficking Machinery: Implications for Oligonucleotide Delivery.Biomedicines. 2021 May 5;9(5):512. doi: 10.3390/biomedicines9050512. Biomedicines. 2021. PMID: 34063104 Free PMC article. Review.
References
-
- Kooijman EE, Chupin V, de Kruijff B, Burger KN. Modulation of membrane curvature by phosphatidic acid and lysophosphatidic acid. Traffic. 2003;4:162–174. - PubMed
-
- Bonifacino JS, Glick BS. The mechanisms of vesicle budding and fusion. Cell. 2004;116:153–166. - PubMed
-
- Lee MC, Miller EA, Goldberg J, Orci L, Schekman R. Bi-directional protein transport between the ER and Golgi. Annu Rev Cell Dev Biol. 2004;20:87–123. - PubMed
-
- Schekman R. Lasker Basic Medical Research Award. SEC mutants and the secretory apparatus. Nat Med. 2002;8:1055–1058. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

