Cdc1p is an endoplasmic reticulum-localized putative lipid phosphatase that affects Golgi inheritance and actin polarization by activating Ca2+ signaling
- PMID: 18332110
- PMCID: PMC2423168
- DOI: 10.1128/MCB.00567-07
Cdc1p is an endoplasmic reticulum-localized putative lipid phosphatase that affects Golgi inheritance and actin polarization by activating Ca2+ signaling
Abstract
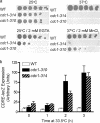
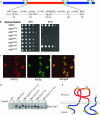
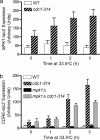
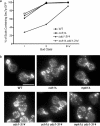
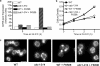
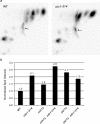
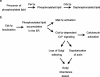
In the budding yeast Saccharomyces cerevisiae, mutations in the essential gene CDC1 cause defects in Golgi inheritance and actin polarization. However, the biochemical function of Cdc1p is unknown. Previous work showed that cdc1 mutants accumulate intracellular Ca(2+) and display enhanced sensitivity to the extracellular Mn(2+) concentration, suggesting that Cdc1p might regulate divalent cation homeostasis. By contrast, our data indicate that Cdc1p is a Mn(2+)-dependent protein that can affect Ca(2+) levels. We identified a cdc1 allele that activates Ca(2+) signaling but does not show enhanced sensitivity to the Mn(2+) concentration. Furthermore, our studies show that Cdc1p is an endoplasmic reticulum-localized transmembrane protein with a putative phosphoesterase domain facing the lumen. cdc1 mutant cells accumulate an unidentified phospholipid, suggesting that Cdc1p may be a lipid phosphatase. Previous work showed that deletion of the plasma membrane Ca(2+) channel Cch1p partially suppressed the cdc1 growth phenotype, and we find that deletion of Cch1p also suppresses the Golgi inheritance and actin polarization phenotypes. The combined data fit a model in which the cdc1 mutant phenotypes result from accumulation of a phosphorylated lipid that activates Ca(2+) signaling.
Figures
Similar articles
-
Cdc1p is a Golgi-localized glycosylphosphatidylinositol-anchored protein remodelase.Mol Biol Cell. 2020 Dec 15;31(26):2883-2891. doi: 10.1091/mbc.E20-08-0539. Epub 2020 Oct 28. Mol Biol Cell. 2020. PMID: 33112703 Free PMC article.
-
A role for actin, Cdc1p, and Myo2p in the inheritance of late Golgi elements in Saccharomyces cerevisiae.J Cell Biol. 2001 Apr 2;153(1):47-62. doi: 10.1083/jcb.153.1.47. J Cell Biol. 2001. PMID: 11285273 Free PMC article.
-
Cdc1 removes the ethanolamine phosphate of the first mannose of GPI anchors and thereby facilitates the integration of GPI proteins into the yeast cell wall.Mol Biol Cell. 2014 Nov 1;25(21):3375-88. doi: 10.1091/mbc.E14-06-1033. Epub 2014 Aug 27. Mol Biol Cell. 2014. PMID: 25165136 Free PMC article.
-
Discoveries of the phosphatidate phosphatase genes in yeast published in the Journal of Biological Chemistry.J Biol Chem. 2019 Feb 1;294(5):1681-1689. doi: 10.1074/jbc.TM118.004159. Epub 2018 Jul 30. J Biol Chem. 2019. PMID: 30061152 Free PMC article. Review.
-
Inheritance of the endoplasmic reticulum and Golgi apparatus.Curr Opin Cell Biol. 2002 Aug;14(4):496-9. doi: 10.1016/s0955-0674(02)00345-9. Curr Opin Cell Biol. 2002. PMID: 12383802 Review.
Cited by
-
Potential Physiological Relevance of ERAD to the Biosynthesis of GPI-Anchored Proteins in Yeast.Int J Mol Sci. 2021 Jan 21;22(3):1061. doi: 10.3390/ijms22031061. Int J Mol Sci. 2021. PMID: 33494405 Free PMC article. Review.
-
The yeast Golgi apparatus: insights and mysteries.FEBS Lett. 2009 Dec 3;583(23):3746-51. doi: 10.1016/j.febslet.2009.10.072. Epub 2009 Oct 29. FEBS Lett. 2009. PMID: 19879270 Free PMC article. Review.
-
The yeast p5 type ATPase, spf1, regulates manganese transport into the endoplasmic reticulum.PLoS One. 2013 Dec 31;8(12):e85519. doi: 10.1371/journal.pone.0085519. eCollection 2013. PLoS One. 2013. PMID: 24392018 Free PMC article.
-
Impaired manganese metabolism causes mitotic misregulation.J Biol Chem. 2012 May 25;287(22):18717-29. doi: 10.1074/jbc.M112.358309. Epub 2012 Apr 4. J Biol Chem. 2012. PMID: 22493290 Free PMC article.
-
Cdc1p is a Golgi-localized glycosylphosphatidylinositol-anchored protein remodelase.Mol Biol Cell. 2020 Dec 15;31(26):2883-2891. doi: 10.1091/mbc.E20-08-0539. Epub 2020 Oct 28. Mol Biol Cell. 2020. PMID: 33112703 Free PMC article.
References
-
- Abeijon, C., and C. B. Hirschberg. 1992. Topography of glycosylation reactions in the endoplasmic reticulum. Trends Biochem. Sci. 1732-36. - PubMed
-
- Ayscough, K. R., J. Stryker, N. Pokala, M. Sanders, P. Crews, and D. G. Drubin. 1997. High rates of actin filament turnover in budding yeast and roles for actin in establishment and maintenance of cell polarity revealed using the actin inhibitor latrunculin-A. J. Cell Biol. 137399-416. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
