Functional characterization of murine interferon regulatory factor 5 (IRF-5) and its role in the innate antiviral response
- PMID: 18332133
- PMCID: PMC2386920
- DOI: 10.1074/jbc.M800501200
Functional characterization of murine interferon regulatory factor 5 (IRF-5) and its role in the innate antiviral response
Abstract
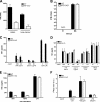
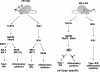
Although the role of human IRF-5 in antiviral and inflammatory responses in vitro has been well characterized, much remains to be elucidated about murine IRF-5. Murine IRF-5, unlike the heavily spliced human gene, is primarily expressed as a full-length transcript, with only a single splice variant that was detected in very low levels in the bone marrow of C57BL/6J mice. This bone marrow variant contains a 288-nucleotide deletion from exons 4-6 and exhibits impaired transcriptional activity. The murine IRF-5 can be activated by both TBK1 and MyD88 to form homodimers and bind to and activate transcription of type I interferon and inflammatory cytokine genes. The importance of IRF-5 in the antiviral and inflammatory response in vivo is highlighted by marked reductions in serum levels of type I interferon and tumor necrosis factor alpha (TNFalpha) in Newcastle disease virus-infected Irf5(-)(/)(-) mice. IRF-5 is critical for TLR3-, TLR4-, and TLR9-dependent induction of TNFalpha in CD11c(+) dendritic cells. In contrast, TLR9, but not TLR3/4-mediated induction of type I IFN transcription, is dependent on IRF-5 in these cells. In addition, IRF-5 regulates TNFalpha but not type I interferon gene transcription in Newcastle disease virus-infected peritoneal macrophages. Altogether, these data reveal the cell type-specific importance of IRF-5 in MyD88-mediated antiviral pathways and the widespread role of IRF-5 in the regulation of inflammatory cytokines.
Figures
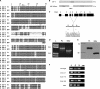
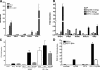
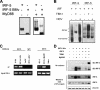
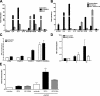
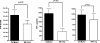
References
-
- Akira, S., Uematsu, S., and Takeuchi, O. (2006) Cell 124 783–801 - PubMed
-
- O'Neill, L. A. (2006) Curr. Opin. Immunol. 18 3–9 - PubMed
-
- Hornung, V., Ellegast, J., Kim, S., Brzozka, K., Jung, A., Kato, H., Poeck, H., Akira, S., Conzelmann, K. K., Schlee, M., Endres, S., and Hartmann, G. (2006) Science 314 994–997 - PubMed
-
- Yoneyama, M., Kikuchi, M., Matsumoto, K., Imaizumi, T., Miyagishi, M., Taira, K., Foy, E., Loo, Y. M., Gale, M., Jr., Akira, S., Yonehara, S., Kato, A., and Fujita, T. (2005) J. Immunol. 175 2851–2858 - PubMed
-
- Yoneyama, M., Kikuchi, M., Natsukawa, T., Shinobu, N., Imaizumi, T., Miyagishi, M., Taira, K., Akira, S., and Fujita, T. (2004) Nat. Immunol. 5 730–737 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

