Ectopic notch activation in developing podocytes causes glomerulosclerosis
- PMID: 18337488
- PMCID: PMC2396929
- DOI: 10.1681/ASN.2007050596
Ectopic notch activation in developing podocytes causes glomerulosclerosis
Abstract
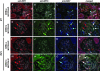
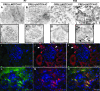
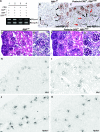
Genetic evidence supports an early role for Notch signaling in the fate of podocytes during glomerular development. Decreased expression of Notch transcriptional targets in developing podocytes after the determination of cell fate suggests that constitutive Notch signaling may oppose podocyte differentiation. This study determined the effects of constitutive Notch signaling on podocyte differentiation by ectopically expressing Notch's intracellular domain (NOTCH-IC), the biologically active, intracellular product of proteolytic cleavage of the Notch receptor, in developing podocytes of transgenic mice. Histologic and molecular analyses revealed normal glomerular morphology and expression of podocyte markers in newborn NOTCH-IC-expressing mice; however, mice developed severe proteinuria and showed evidence of progressive glomerulosclerosis at 2 wk after birth. Features of mature podocytes were lost: Foot processes were effaced; expression of Wt1, Nphs1, and Nphs2 was downregulated; cell-cycle re-entry was induced; and the expression of Pax2 was increased. In contrast, mice with podocyte-specific inactivation of Rbpsuh, which encodes a protein essential for canonical Notch signaling, seemed normal. In addition, the damaging effects of NOTCH-IC expression were prevented in transgenic mice after simultaneous conditional inactivation of Rbpsuh in murine podocytes. These results suggest that Notch signaling is dispensable during terminal differentiation of podocytes but that constitutive (or inappropriate) Notch signaling is deleterious, leading to glomerulosclerosis.
Figures
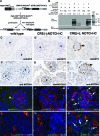
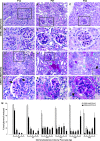

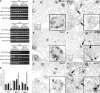
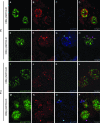

Comment in
-
Notch signaling: a common pathway of injury in podocytopathies?J Am Soc Nephrol. 2008 Jun;19(6):1045-6. doi: 10.1681/ASN.2008040351. Epub 2008 May 14. J Am Soc Nephrol. 2008. PMID: 18480311 No abstract available.
References
-
- Wang P, Pereira FA, Beasley D, Zheng H: Presenilins are required for the formation of comma- and S-shaped bodies during nephrogenesis. Development 130: 5019–5029, 2003 - PubMed
-
- Cheng HT, Miner JH, Lin M, Tansey MG, Roth K, Kopan R: Gamma-secretase activity is dispensable for mesenchyme-to-epithelium transition but required for podocyte and proximal tubule formation in developing mouse kidney. Development 130: 5031–5042, 2003 - PubMed
-
- Kadesch T: Notch signaling: The demise of elegant simplicity. Curr Opin Genet Dev 14: 506–512, 2004 - PubMed
-
- Schroeter EH, Kisslinger JA, Kopan R: Notch-1 signaling requires ligand-induced proteolytic release of intracellular domain. Nature 393: 382–386, 1998 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

