Both MHC class II and its GPI-anchored form undergo hop diffusion as observed by single-molecule tracking
- PMID: 18339737
- PMCID: PMC2426619
- DOI: 10.1529/biophysj.107.123018
Both MHC class II and its GPI-anchored form undergo hop diffusion as observed by single-molecule tracking
Abstract
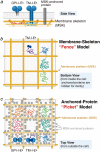
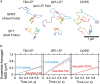
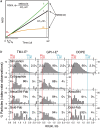
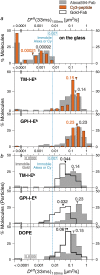
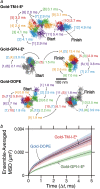
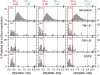
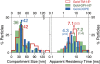
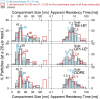
Previously, investigations using single-fluorescent-molecule tracking at frame rates of up to 65 Hz, showed that the transmembrane MHC class II protein and its GPI-anchored modified form expressed in CHO cells undergo simple Brownian diffusion, without any influence of actin depolymerization with cytochalasin D. These results are at apparent variance with the view that GPI-anchored proteins stay with cholesterol-enriched raft domains, as well as with the observation that both lipids and transmembrane proteins undergo short-term confined diffusion within a compartment and long-term hop diffusion between compartments. Here, this apparent discrepancy has been resolved by reexamining the same paradigm, by using both high-speed single-particle tracking (50 kHz) and single fluorescent-molecule tracking (30 Hz). Both molecules exhibited rapid hop diffusion between 40-nm compartments, with an average dwell time of 1-3 ms in each compartment. Cytochalasin D hardly affected the hop diffusion, consistent with previous observations, whereas latrunculin A increased the compartment sizes with concomitant decreases of the hop rates, which led to an approximately 50% increase in the median macroscopic diffusion coefficient. These results indicate that the actin-based membrane skeleton influences the diffusion of both transmembrane and GPI-anchored proteins.
Figures

Similar articles
-
Dynamic Meso-Scale Anchorage of GPI-Anchored Receptors in the Plasma Membrane: Prion Protein vs. Thy1.Cell Biochem Biophys. 2017 Dec;75(3-4):399-412. doi: 10.1007/s12013-017-0808-3. Epub 2017 Jun 24. Cell Biochem Biophys. 2017. PMID: 28646414 Free PMC article.
-
(Un)confined diffusion of CD59 in the plasma membrane determined by high-resolution single molecule microscopy.Biophys J. 2007 May 15;92(10):3719-28. doi: 10.1529/biophysj.106.095398. Epub 2007 Feb 26. Biophys J. 2007. PMID: 17325009 Free PMC article.
-
Sphingomyelin chain length influences the distribution of GPI-anchored proteins in rafts in supported lipid bilayers.Mol Membr Biol. 2007 May-Jun;24(3):233-42. doi: 10.1080/09687860601127770. Mol Membr Biol. 2007. PMID: 17520480
-
Biosynthesis, remodelling and functions of mammalian GPI-anchored proteins: recent progress.J Biochem. 2008 Sep;144(3):287-94. doi: 10.1093/jb/mvn090. Epub 2008 Jul 17. J Biochem. 2008. PMID: 18635593 Review.
-
Paradigm shift of the plasma membrane concept from the two-dimensional continuum fluid to the partitioned fluid: high-speed single-molecule tracking of membrane molecules.Annu Rev Biophys Biomol Struct. 2005;34:351-78. doi: 10.1146/annurev.biophys.34.040204.144637. Annu Rev Biophys Biomol Struct. 2005. PMID: 15869394 Review.
Cited by
-
Lateral membrane diffusion modulated by a minimal actin cortex.Biophys J. 2013 Apr 2;104(7):1465-75. doi: 10.1016/j.bpj.2013.02.042. Biophys J. 2013. PMID: 23561523 Free PMC article.
-
Understanding immune signaling using advanced imaging techniques.Biochem Soc Trans. 2022 Apr 29;50(2):853-866. doi: 10.1042/BST20210479. Biochem Soc Trans. 2022. PMID: 35343569 Free PMC article. Review.
-
Oxygen depletion speeds and simplifies diffusion in HeLa cells.Biophys J. 2014 Oct 21;107(8):1873-1884. doi: 10.1016/j.bpj.2014.08.023. Biophys J. 2014. PMID: 25418168 Free PMC article.
-
A Simple and Powerful Analysis of Lateral Subdiffusion Using Single Particle Tracking.Biophys J. 2017 Dec 5;113(11):2452-2463. doi: 10.1016/j.bpj.2017.09.017. Biophys J. 2017. PMID: 29211999 Free PMC article.
-
Tracking microdomain dynamics in cell membranes.Biochim Biophys Acta. 2009 Jan;1788(1):245-53. doi: 10.1016/j.bbamem.2008.10.024. Epub 2008 Nov 11. Biochim Biophys Acta. 2009. PMID: 19041847 Free PMC article. Review.
References
-
- Kusumi, A., and Y. Sako. 1996. Cell surface organization by the membrane skeleton. Curr. Opin. Cell Biol. 8:566–574. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

