Cytochrome bc1-cy fusion complexes reveal the distance constraints for functional electron transfer between photosynthesis components
- PMID: 18343816
- PMCID: PMC2376229
- DOI: 10.1074/jbc.M800091200
Cytochrome bc1-cy fusion complexes reveal the distance constraints for functional electron transfer between photosynthesis components
Abstract
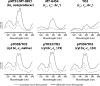
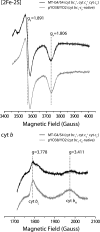
Photosynthetic (Ps) growth of purple non-sulfur bacteria such as Rhodobacter capsulatus depends on the cyclic electron transfer (ET) between the ubihydroquinone (QH2): cytochrome (cyt) c oxidoreductases (cyt bc1 complex), and the photochemical reaction centers (RC), mediated by either a membrane-bound (cyt c(y)) or a freely diffusible (cyt c2) electron carrier. Previously, we constructed a functional cyt bc1-c(y) fusion complex that supported Ps growth solely relying on membrane-confined ET ( Lee, D.-W., Ozturk, Y., Mamedova, A., Osyczka, A., Cooley, J. W., and Daldal, F. (2006) Biochim. Biophys. Acta 1757, 346-352 ). In this work, we further characterized this cyt bc1-c(y) fusion complex, and used its derivatives with shorter cyt c(y) linkers as "molecular rulers" to probe the distances separating the Ps components. Comparison of the physicochemical properties of both membrane-embedded and purified cyt bc1-c(y) fusion complexes established that these enzymes were matured and assembled properly. Light-activated, time-resolved kinetic spectroscopy analyses revealed that their variants with shorter cyt c(y) linkers exhibited fast, native-like ET rates to the RC via the cyt bc1. However, shortening the length of the cyt c(y) linker decreased drastically this electronic coupling between the cyt bc1-c(y) fusion complexes and the RC, thereby limiting Ps growth. The shortest and still functional cyt c(y) linker was about 45 amino acids long, showing that the minimal distance allowed between the cyt bc1-c(y) fusion complexes and the RC and their surrounding light harvesting proteins was very short. These findings support the notion that membrane-bound Ps components form large, active structural complexes that are "hardwired" for cyclic ET.
Figures





Similar articles
-
A functional hybrid between the cytochrome bc1 complex and its physiological membrane-anchored electron acceptor cytochrome cy in Rhodobacter capsulatus.Biochim Biophys Acta. 2006 May-Jun;1757(5-6):346-52. doi: 10.1016/j.bbabio.2006.04.025. Epub 2006 May 9. Biochim Biophys Acta. 2006. PMID: 16781662
-
Soluble variants of Rhodobacter capsulatus membrane-anchored cytochrome cy are efficient photosynthetic electron carriers.J Biol Chem. 2008 May 16;283(20):13964-72. doi: 10.1074/jbc.M800090200. Epub 2008 Mar 14. J Biol Chem. 2008. PMID: 18343817 Free PMC article.
-
Membrane-anchored cytochrome cy mediated microsecond time range electron transfer from the cytochrome bc1 complex to the reaction center in Rhodobacter capsulatus.Biochemistry. 1998 Apr 21;37(16):5501-10. doi: 10.1021/bi973123d. Biochemistry. 1998. PMID: 9548933
-
The bc1 complexes of Rhodobacter sphaeroides and Rhodobacter capsulatus.J Bioenerg Biomembr. 1993 Jun;25(3):195-209. doi: 10.1007/BF00762582. J Bioenerg Biomembr. 1993. PMID: 8394316 Review.
-
Structural and functional proteomics of intracytoplasmic membrane assembly in Rhodobacter sphaeroides.J Mol Microbiol Biotechnol. 2013;23(1-2):48-62. doi: 10.1159/000346520. Epub 2013 Apr 18. J Mol Microbiol Biotechnol. 2013. PMID: 23615195 Review.
Cited by
-
Overexpression, characterization, and crystallization of the functional domain of cytochrome c(z) from Chlorobium tepidum.Photosynth Res. 2009 Oct;102(1):77-84. doi: 10.1007/s11120-009-9492-5. Photosynth Res. 2009. PMID: 19731072
-
Intermonomer electron transfer between the low-potential b hemes of cytochrome bc₁.Biochemistry. 2011 Mar 15;50(10):1651-63. doi: 10.1021/bi101736v. Epub 2011 Feb 15. Biochemistry. 2011. PMID: 21261281 Free PMC article.
-
Catalytically-relevant electron transfer between two hemes bL in the hybrid cytochrome bc1-like complex containing a fusion of Rhodobacter sphaeroides and capsulatus cytochromes b.Biochim Biophys Acta. 2013 Jun;1827(6):751-60. doi: 10.1016/j.bbabio.2013.02.007. Epub 2013 Feb 18. Biochim Biophys Acta. 2013. PMID: 23428397 Free PMC article.
-
Molecular mechanisms of superoxide production by complex III: a bacterial versus human mitochondrial comparative case study.Biochim Biophys Acta. 2013 Nov-Dec;1827(11-12):1332-9. doi: 10.1016/j.bbabio.2013.03.009. Epub 2013 Mar 28. Biochim Biophys Acta. 2013. PMID: 23542447 Free PMC article. Review.
-
The CopA2-Type P1B-Type ATPase CcoI Serves as Central Hub for cbb 3-Type Cytochrome Oxidase Biogenesis.Front Microbiol. 2021 Sep 13;12:712465. doi: 10.3389/fmicb.2021.712465. eCollection 2021. Front Microbiol. 2021. PMID: 34589071 Free PMC article.
References
-
- Berry, E. A., Guergova-Kuras, M., Huang, L. S., and Crofts, A. R. (2000) Annu. Rev. Biochem. 691005 -1075 - PubMed
-
- Darrouzet, E., Cooley, J. W., and Daldal, F. (2004) Photosynth. Res. 7925 -44 - PubMed
-
- Crofts, A. R., and Meinhardt, S. W. (1982) Biochem. Soc. Trans. 10201 -203 - PubMed
-
- Gennis, R. B., Barquera, B., Hacker, B., Van Doren, S. R., Arnaud, S., Crofts, A. R., Davidson, E., Gray, K. A., and Daldal, F. (1993) J. Bioenerg. Biomembr. 25 195-209 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

