Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment
- PMID: 18348728
- PMCID: PMC2373291
- DOI: 10.1186/1742-2094-5-10
Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment
Abstract
Background: Cytokines and alcohol share a common modulation of inflammation and hormones as well as being implicated in multiple diseases, but the mechanisms are poorly understood. The purpose of this study was to investigate the interaction of liver, serum and brain cytokines as well as whether ethanol would potentiate endotoxin (Lipopolysaccharide, LPS) responses once ethanol had cleared.
Methods: Male C57BL/6J mice were treated intragastrically with water (control) or ethanol (5 g/kg, i.g., 25% ethanol, w/v), with volumes matched, for 1 day or daily for 10 days. Mice were then injected intraperitoneally with saline (control) or LPS (3 mg/kg, i.p.) in saline 24 hrs after the last dose of ethanol. Gene expression and protein synthesis of proinflammatory cytokines and anti-inflammatory cytokine, oxidative enzymes, microglial activation and inhibition of neurogenesis were examined using real-time PCR, ELISA, and immunohistochemistry.
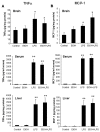
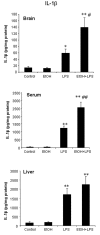
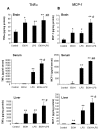
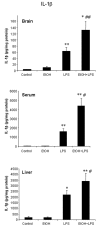
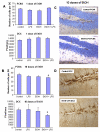
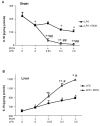
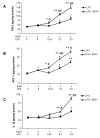
Results: LPS increased proinflammatory cytokines (TNFalpha, MCP-1, IL-1beta) several fold in liver, brain and serum at 1 hr. Ethanol is known to increase liver cytokines and alter the risk of multiple chronic diseases. Ten daily doses of ethanol increased brain and liver TNFalpha, and pretreatment with ethanol potentiated LPS-induced increases in TNFalpha, MCP-1, IL-1beta in liver, serum and brain. Proinflammatory cytokine levels in liver and serum returned to basal levels within a day, whereas brain proinflammatory cytokines remained elevated for long periods. IL-10, an anti-inflammatory cytokine, is reduced in brain by ethanol and LPS, while brain proinflammatory cytokines remain increased, whereas liver IL-10 is increased when proinflammatory cytokines have returned to control levels. Activation of brain microglia indicated by morphological changes, reduced neurogenesis and increased brain expression of COX-2 and gp91phox NADPH oxidase subunit mRNA were found in the 10 daily doses of ethanol-pretreated LPS group.
Conclusion: Acute increases in serum cytokines induce long lasting increases in brain proinflammatory cytokines. Ten daily doses of ethanol exposure results in persistent alterations of cytokines and significantly increases the magnitude and duration of central and peripheral proinflammatory cytokines and microglial activation. Ethanol induced differential anti-inflammatory cytokine IL-10 responses in liver and brain could cause long lasting disruption of cytokine cascades that could contribute to protection or increased risk of multiple chronic diseases.
Figures

Similar articles
-
Reduced gut microbiome protects from alcohol-induced neuroinflammation and alters intestinal and brain inflammasome expression.J Neuroinflammation. 2018 Oct 27;15(1):298. doi: 10.1186/s12974-018-1328-9. J Neuroinflammation. 2018. PMID: 30368255 Free PMC article.
-
Ethanol-induced sensitization to endotoxin in Kupffer cells is dependent upon oxidative stress.Alcohol Clin Exp Res. 2005 Dec;29(12 Suppl):246S-50S. doi: 10.1097/01.alc.0000191128.54871.40. Alcohol Clin Exp Res. 2005. PMID: 16385231
-
Chronic ethanol increases systemic TLR3 agonist-induced neuroinflammation and neurodegeneration.J Neuroinflammation. 2012 Jun 18;9:130. doi: 10.1186/1742-2094-9-130. J Neuroinflammation. 2012. PMID: 22709825 Free PMC article.
-
Converging actions of alcohol on liver and brain immune signaling.Int Rev Neurobiol. 2014;118:359-80. doi: 10.1016/B978-0-12-801284-0.00011-7. Int Rev Neurobiol. 2014. PMID: 25175869 Review.
-
Neuroinflammation as a neurotoxic mechanism in alcoholism: commentary on "Increased MCP-1 and microglia in various regions of human alcoholic brain".Exp Neurol. 2008 Sep;213(1):10-7. doi: 10.1016/j.expneurol.2008.05.016. Epub 2008 Jul 14. Exp Neurol. 2008. PMID: 18625499 Free PMC article. Review. No abstract available.
Cited by
-
Daily exercise improves memory, stimulates hippocampal neurogenesis and modulates immune and neuroimmune cytokines in aging rats.Brain Behav Immun. 2013 Feb;28:25-43. doi: 10.1016/j.bbi.2012.09.013. Epub 2012 Oct 16. Brain Behav Immun. 2013. PMID: 23078985 Free PMC article.
-
Chronic ethanol exposure combined with high fat diet up-regulates P2X7 receptors that parallels neuroinflammation and neuronal loss in C57BL/6J mice.J Neuroimmunol. 2015 Aug 15;285:169-79. doi: 10.1016/j.jneuroim.2015.06.007. Epub 2015 Jun 20. J Neuroimmunol. 2015. PMID: 26198936 Free PMC article.
-
NADPH oxidase and reactive oxygen species contribute to alcohol-induced microglial activation and neurodegeneration.J Neuroinflammation. 2012 Jan 12;9:5. doi: 10.1186/1742-2094-9-5. J Neuroinflammation. 2012. PMID: 22240163 Free PMC article.
-
Inflammatory responses to alcohol in the CNS: nuclear receptors as potential therapeutics for alcohol-induced neuropathologies.J Leukoc Biol. 2016 Nov;100(5):951-959. doi: 10.1189/jlb.3MR0416-171R. Epub 2016 Jul 26. J Leukoc Biol. 2016. PMID: 27462100 Free PMC article. Review.
-
A dialogue between the immune system and brain, spoken in the language of serotonin.ACS Chem Neurosci. 2013 Jan 16;4(1):48-63. doi: 10.1021/cn300186b. Epub 2012 Dec 7. ACS Chem Neurosci. 2013. PMID: 23336044 Free PMC article. Review.
References
-
- Simi A, Tsakiri N, Wang P, Rothwell NJ. Interleukin-1 and inflammatory neurodegeneration. Biochem Soc Trans. 2007;35:1122–1126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

