The role of chloride anion and CFTR in killing of Pseudomonas aeruginosa by normal and CF neutrophils
- PMID: 18353929
- PMCID: PMC2901559
- DOI: 10.1189/jlb.0907658
The role of chloride anion and CFTR in killing of Pseudomonas aeruginosa by normal and CF neutrophils
Abstract
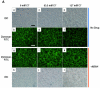
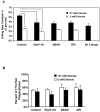
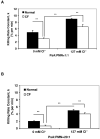
Chloride anion is essential for myeloperoxidase (MPO) to produce hypochlorous acid (HOCl) in polymorphonuclear neutrophils (PMNs). To define whether chloride availability to PMNs affects their HOCl production and microbicidal capacity, we examined how extracellular chloride concentration affects killing of Pseudomonas aeruginosa (PsA) by normal neutrophils. PMN-mediated bacterial killing was strongly dependent on extracellular chloride concentration. Neutrophils in a chloride-deficient medium killed PsA poorly. However, as the chloride level was raised, the killing efficiency increased in a dose-dependent manner. By using specific inhibitors to selectively block NADPH oxidase, MPO, and cystic fibrosis transmembrane conductance regulator (CFTR) functions, neutrophil-mediated killing of PsA could be attributed to three distinct mechanisms: CFTR-dependent and oxidant-dependent; chloride-dependent but not CFTR- and oxidant-dependent; and independent of any of the tested factors. Therefore, chloride anion is involved in oxidant- and nonoxidant-mediated bacterial killing. We previously reported that neutrophils from CF patients are defective in chlorination of ingested bacteria, suggesting that the chloride channel defect might impair the MPO-hydrogen peroxide-chloride microbicidal function. Here, we compared the competence of killing PsA by neutrophils from normal donors and CF patients. The data demonstrate that the killing rate by CF neutrophils was significantly lower than that by normal neutrophils. CF neutrophils in a chloride-deficient environment had only one-third of the bactericidal capacity of normal neutrophils in a physiological chloride environment. These results suggest that CFTR-dependent chloride anion transport contributes significantly to killing PsA by normal neutrophils and when defective as in CF, may compromise the ability to clear PsA.
Figures



References
-
- Klebanoff SJ. Myeloperoxidase: friend and foe. J. Leukoc. Biol. 2005;77:598–625. - PubMed
-
- Ganz T, Lehrer RI. Defensins. Pharmacol Ther. 1995;66:191–205. - PubMed
-
- Hampton MB, Kettle AJ, Winterbourn CC. Inside the neutrophil phagosome: oxidants, myeloperoxidase, and bacterial killing. Blood. 1998;92:3007–17. - PubMed
-
- Sbarra AJ, Karnovsky ML. The biochemical basis of phagocytosis. I. Metabolic changes during the ingestion of particles by polymorphonuclear leukocytes. J Biol Chem. 1959;234(6):1355–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

