Variant-specific [PSI+] infection is transmitted by Sup35 polymers within [PSI+] aggregates with heterogeneous protein composition
- PMID: 18353968
- PMCID: PMC2397312
- DOI: 10.1091/mbc.e08-01-0078
Variant-specific [PSI+] infection is transmitted by Sup35 polymers within [PSI+] aggregates with heterogeneous protein composition
Abstract
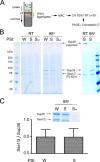
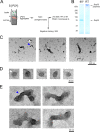
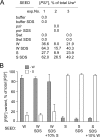
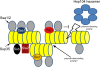
The [PSI(+)] prion is the aggregated self-propagating form of the Sup35 protein from the yeast Saccharomyces cerevisiae. Aggregates of Sup35 in [PSI(+)] cells exist in different heritable conformations, called "variants," and they are composed of detergent-resistant Sup35 polymers, which may be closely associated with themselves, other proteins, or both. Here, we report that disassembly of the aggregates into individual Sup35 polymers and non-Sup35 components increases their infectivity while retaining their variant specificity, showing that variant-specific [PSI(+)] infection can be transmitted by Sup35 polymers alone. Morphological analysis revealed that Sup35 isolated from [PSI(+)] yeast has the appearance of short barrels, and bundles, which seem to be composed of barrels. We show that the major components of two different variants of [PSI(+)] are interacting infectious Sup35 polymers and Ssa1/2. Using a candidate approach, we detected Hsp104, Ssb1/2, Sis1, Sse1, Ydj1, and Sla2 among minor components of the aggregates. We demonstrate that Ssa1/2 efficiently binds to the prion domain of Sup35 in [PSI(+)] cells, but that it interacts poorly with the nonaggregated Sup35 found in [psi(-)] cells. Hsp104, Sis1, and Sse1 interact preferentially with the prion versus nonprion form of Sup35, whereas Sla2 and Ssb1/2 interact with both forms of Sup35 with similar efficiency.
Figures



References
-
- Allen K. D., Chernova T. A., Tennant E. P., Wilkinson K. D., Chernoff Y. O. Effects of ubiquitin system alterations on the formation and loss of a yeast prion. J. Biol. Chem. 2007;282:3004–3013. - PubMed
-
- Allen K. D., Wegrzyn R. D., Chernova T. A., Muller S., Newnam G. P., Winslett P. A., Wittich K. B., Wilkinson K. D., Chernoff Y. O. Hsp70 chaperones as modulators of prion life cycle: novel effects of Ssa and Ssb on the Saccharomyces cerevisiae prion [PSI+] Genetics. 2005;169:1227–1242. - PMC - PubMed
-
- Bagriantsev S., Liebman S. W. Specificity of prion assembly in vivo: [PSI+] and [PIN+] form separate structures in yeast. J. Biol. Chem. 2004;279:51042–51048. - PubMed
-
- Bagriantsev S. N., Kushnirov V. V., Liebman S. W. Analysis of amyloid aggregates using agarose gel electrophoresis. Methods Enzymol. 2006;412:33–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

