Synaptic metaplasticity through NMDA receptor lateral diffusion
- PMID: 18354009
- PMCID: PMC6670713
- DOI: 10.1523/JNEUROSCI.5450-07.2008
Synaptic metaplasticity through NMDA receptor lateral diffusion
Retraction in
-
Retraction. Synaptic metaplasticity through NMDA receptor lateral diffusion.J Neurosci. 2011 Jun 22;31(25):9441. J Neurosci. 2011. PMID: 21697393 Free PMC article. No abstract available.
Abstract
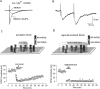
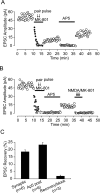
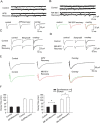
Lateral diffusion of glutamate receptors was proposed as a mechanism for regulating receptor numbers at synapses and affecting synaptic functions, especially the efficiency of synaptic transmission. However, a direct link between receptor lateral diffusion and change in synaptic function has not yet been established. In the present study, we demonstrated NMDA receptor (NMDAR) lateral diffusion in CA1 neurons in hippocampal slices by detecting considerable recovery of spontaneous or evoked EPSCs from the block of (+)-MK-801 [(+)-5-methyl-10,11-dihydro-5H-dibenzo [a,d] cyclohepten-5,10-imine maleate], an irreversible NMDAR open-channel blocker. We observed changes on both the number and the composition of synaptic NMDAR on recovery. More importantly, after the recovery, long-term potentiation (LTP)-producing protocol induced only LTD (long-term depression) instead of LTP. In contrast, a complete recovery from competitive NMDAR blocker D,L-AP-5 was observed without subsequent changes on synaptic plasticity. Our data suggest a revised model of NMDAR trafficking wherein extrasynaptic NMDARs, mostly NR1/NR2B receptors, move laterally into synaptic sites, resulting in altered rule of synaptic modification. Thus, CA1 synapses exhibit a novel form of metaplasticity in which the direction of synaptic modification can be reverted through subtype-specific lateral diffusion of NMDA receptors.
Figures



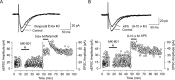

Similar articles
-
Extrasynaptic NR2D-containing NMDARs are recruited to the synapse during LTP of NMDAR-EPSCs.J Neurosci. 2008 Nov 5;28(45):11685-94. doi: 10.1523/JNEUROSCI.3035-08.2008. J Neurosci. 2008. PMID: 18987204 Free PMC article.
-
Mechanisms of group I mGluR-dependent long-term depression of NMDA receptor-mediated transmission at Schaffer collateral-CA1 synapses.J Neurophysiol. 2009 Mar;101(3):1375-85. doi: 10.1152/jn.90643.2008. Epub 2008 Dec 24. J Neurophysiol. 2009. PMID: 19109458
-
N-methyl-D-aspartate receptor-dependent long-term potentiation in CA1 region affects synaptic expression of glutamate receptor subunits and associated proteins in the whole hippocampus.Neuroscience. 2006 Sep 1;141(3):1399-413. doi: 10.1016/j.neuroscience.2006.04.070. Epub 2006 Jun 12. Neuroscience. 2006. PMID: 16766131
-
Hippocampal long-term synaptic plasticity and signal amplification of NMDA receptors.Crit Rev Neurobiol. 2006;18(1-2):71-84. doi: 10.1615/critrevneurobiol.v18.i1-2.80. Crit Rev Neurobiol. 2006. PMID: 17725510 Review.
-
NMDA receptors and synaptic plasticity in the anterior cingulate cortex.Neuropharmacology. 2021 Oct 1;197:108749. doi: 10.1016/j.neuropharm.2021.108749. Epub 2021 Aug 5. Neuropharmacology. 2021. PMID: 34364898 Review.
Cited by
-
NR2A and NR2B subunits differentially mediate MAP kinase signaling and mitochondrial morphology following excitotoxic insult.Neurochem Int. 2012 Apr;60(5):506-16. doi: 10.1016/j.neuint.2012.02.007. Epub 2012 Feb 15. Neurochem Int. 2012. PMID: 22366650 Free PMC article.
-
The Effects of Poria cocos on Rho Signaling-Induced Regulation of Mobility and F-Actin Aggregation in MK-801-Treated B35 and C6 Cells.Behav Neurol. 2022 Jul 12;2022:8225499. doi: 10.1155/2022/8225499. eCollection 2022. Behav Neurol. 2022. PMID: 35864844 Free PMC article.
-
Glutamate receptor dynamics in dendritic microdomains.Neuron. 2008 May 22;58(4):472-97. doi: 10.1016/j.neuron.2008.04.030. Neuron. 2008. PMID: 18498731 Free PMC article. Review.
-
Mobility of NMDA autoreceptors but not postsynaptic receptors at glutamate synapses in the rat entorhinal cortex.J Physiol. 2008 Oct 15;586(20):4905-24. doi: 10.1113/jphysiol.2008.157974. Epub 2008 Aug 21. J Physiol. 2008. PMID: 18718983 Free PMC article.
-
Dynamic and specific interaction between synaptic NR2-NMDA receptor and PDZ proteins.Proc Natl Acad Sci U S A. 2010 Nov 9;107(45):19561-6. doi: 10.1073/pnas.1002690107. Epub 2010 Oct 25. Proc Natl Acad Sci U S A. 2010. PMID: 20974938 Free PMC article.
References
-
- Abraham WC, Bear MF. Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci. 1996;19:126–130. - PubMed
-
- Adesnik H, Nicoll RA, England PM. Photoinactivation of native AMPA receptors reveals their real-time trafficking. Neuron. 2005;48:977–985. - PubMed
-
- Bartlett TE, Bannister NJ, Collett VJ, Dargan SL, Massey PV, Bortolotto ZA, Fitzjohn SM, Bashir ZI, Collingridge GL, Lodge D. Differential roles of NR2A and NR2B-containing NMDA receptors in LTP and LTD in the CA1 region of two-week old rat hippocampus. Neuropharmacology. 2007;52:60–70. - PubMed
-
- Bekkers JM, Stevens CF. Presynaptic mechanism for long-term potentiation in the hippocampus. Nature. 1990;346:724–729. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
