Spinal microglial and perivascular cell cannabinoid receptor type 2 activation reduces behavioral hypersensitivity without tolerance after peripheral nerve injury
- PMID: 18362605
- PMCID: PMC2647363
- DOI: 10.1097/ALN.0b013e318167af74
Spinal microglial and perivascular cell cannabinoid receptor type 2 activation reduces behavioral hypersensitivity without tolerance after peripheral nerve injury
Abstract
Background: Cannabinoids induce analgesia by acting on cannabinoid receptor (CBR) types 1 and/or 2. However, central nervous system side effects and antinociceptive tolerance from CBR1 limit their clinical use. CBR2 exist on spinal glia and perivascular cells, suggesting an immunoregulatory role of these receptors in the central nervous system. Previously, the authors showed that spinal CBR2 activation reduces paw incision hypersensitivity and glial activation. This study tested whether CBR2 are expressed in glia and whether their activation would induce antinociception, glial inhibition, central side effects, and antinociceptive tolerance in a neuropathic rodent pain model.
Methods: Rats underwent L5 spinal nerve transection or sham surgery, and CBR2 expression and cell localization were assessed by immunohistochemistry. Animals received intrathecal injections of CBR agonists and antagonists, and mechanical withdrawal thresholds and behavioral side effects were assessed.
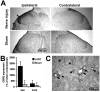
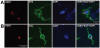
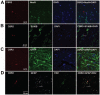
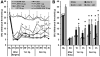
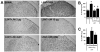
Results: Peripheral nerve transection induced hypersensitivity, increased expression of CR3/CD11b and CBR2, and reduced ED2/CD163 expression in the spinal cord. The CBR2 were localized to microglia and perivascular cells. Intrathecal JWH015 reduced peripheral nerve injury hypersensitivity and CR3/CD11b expression and increased ED2/CD163 expression in a dose-dependent fashion. These effects were prevented by intrathecal administration of the CBR2 antagonist (AM630) but not the CBR1 antagonist (AM281). JWH015 did not cause behavioral side effects. Chronic intrathecal JWH015 treatment did not induce antinociceptive tolerance.
Conclusions: These data indicate that intrathecal CBR2 agonists may provide analgesia by modulating the spinal immune response and microglial function in chronic pain conditions without inducing tolerance and neurologic side effects.
Figures




References
-
- Dworkin RH, Backonja M, Rowbotham MC, Allen RR, Argoff CR, Bennett GJ, Bushnell MC, Farrar JT, Galer BS, Haythornthwaite JA, Hewitt DJ, Loeser JD, Max MB, Saltarelli M, Schmader KE, Stein C, Thompson D, Turk DC, Wallace MS, Watkins LR, Weinstein SM. Advances in neuropathic pain: Diagnosis, mechanisms, and treatment recommendations. Arch Neurol. 2003;60:1524–34. - PubMed
-
- Bridges D, Rice AS, Egertova M, Elphick MR, Winter J, Michael GJ. Localisation of cannabinoid receptor 1 in rat dorsal root ganglion using in situ hybridisation and immunohistochemistry. Neuroscience. 2003;119:803–12. - PubMed
-
- Farquhar-Smith WP, Egertova M, Bradbury EJ, McMahon SB, Rice AS, Elphick MR. Cannabinoid CB(1) receptor expression in rat spinal cord. Mol Cell Neurosci. 2000;15:510–21. - PubMed
-
- Lim G, Sung B, Ji RR, Mao J. Upregulation of spinal cannabinoid-1-receptors following nerve injury enhances the effects of Win 55,212-2 on neuropathic pain behaviors in rats. Pain. 2003;105:275–83. - PubMed
-
- Pettit DA, Harrison MP, Olson JM, Spencer RF, Cabral GA. Immunohistochemical localization of the neural cannabinoid receptor in rat brain. J Neurosci Res. 1998;51:391–402. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

