Alpha-tocopheryl succinate induces apoptosis by targeting ubiquinone-binding sites in mitochondrial respiratory complex II
- PMID: 18372923
- PMCID: PMC2668987
- DOI: 10.1038/onc.2008.69
Alpha-tocopheryl succinate induces apoptosis by targeting ubiquinone-binding sites in mitochondrial respiratory complex II
Abstract
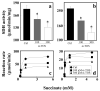
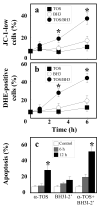
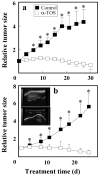
Alpha-tocopheryl succinate (alpha-TOS) is a selective inducer of apoptosis in cancer cells, which involves the accumulation of reactive oxygen species (ROS). The molecular target of alpha-TOS has not been identified. Here, we show that alpha-TOS inhibits succinate dehydrogenase (SDH) activity of complex II (CII) by interacting with the proximal and distal ubiquinone (UbQ)-binding site (Q(P) and Q(D), respectively). This is based on biochemical analyses and molecular modelling, revealing similar or stronger interaction energy of alpha-TOS compared to that of UbQ for the Q(P) and Q(D) sites, respectively. CybL-mutant cells with dysfunctional CII failed to accumulate ROS and underwent apoptosis in the presence of alpha-TOS. Similar resistance was observed when CybL was knocked down with siRNA. Reconstitution of functional CII rendered CybL-mutant cells susceptible to alpha-TOS. We propose that alpha-TOS displaces UbQ in CII causing electrons generated by SDH to recombine with molecular oxygen to yield ROS. Our data highlight CII, a known tumour suppressor, as a novel target for cancer therapy.
Figures





Similar articles
-
Affinity of vitamin E analogues for the ubiquinone complex II site correlates with their toxicity to cancer cells.Mol Nutr Food Res. 2011 Oct;55(10):1543-51. doi: 10.1002/mnfr.201100066. Epub 2011 Jun 8. Mol Nutr Food Res. 2011. PMID: 21656671
-
Mitochondrial targeting of vitamin E succinate enhances its pro-apoptotic and anti-cancer activity via mitochondrial complex II.J Biol Chem. 2011 Feb 4;286(5):3717-28. doi: 10.1074/jbc.M110.186643. Epub 2010 Nov 8. J Biol Chem. 2011. PMID: 21059645 Free PMC article.
-
Tocopheramine succinate and tocopheryl succinate: mechanism of mitochondrial inhibition and superoxide radical production.Bioorg Med Chem. 2014 Jan 15;22(2):684-91. doi: 10.1016/j.bmc.2013.12.036. Epub 2013 Dec 25. Bioorg Med Chem. 2014. PMID: 24393721
-
Mitocans as anti-cancer agents targeting mitochondria: lessons from studies with vitamin E analogues, inhibitors of complex II.J Bioenerg Biomembr. 2007 Feb;39(1):65-72. doi: 10.1007/s10863-006-9060-z. J Bioenerg Biomembr. 2007. PMID: 17294131 Review.
-
Vitamin E analogues as inducers of apoptosis: implications for their potential antineoplastic role.Redox Rep. 2001;6(3):143-51. doi: 10.1179/135100001101536247. Redox Rep. 2001. PMID: 11523588 Review.
Cited by
-
The mechanisms of action of mitochondrial targeting agents in cancer: inhibiting oxidative phosphorylation and inducing apoptosis.Front Pharmacol. 2023 Oct 25;14:1243613. doi: 10.3389/fphar.2023.1243613. eCollection 2023. Front Pharmacol. 2023. PMID: 37954849 Free PMC article. Review.
-
Selective Disruption of Respiratory Supercomplexes as a New Strategy to Suppress Her2high Breast Cancer.Antioxid Redox Signal. 2017 Jan 10;26(2):84-103. doi: 10.1089/ars.2016.6677. Epub 2016 Aug 22. Antioxid Redox Signal. 2017. PMID: 27392540 Free PMC article.
-
Targeting mitochondria by α-tocopheryl succinate overcomes hypoxia-mediated tumor cell resistance to treatment.Cell Mol Life Sci. 2014 Jun;71(12):2325-33. doi: 10.1007/s00018-013-1489-8. Epub 2013 Oct 19. Cell Mol Life Sci. 2014. PMID: 24142346 Free PMC article.
-
Multifunctional decoration of alpha-tocopheryl succinate-based NP for cancer treatment: effect of TPP and LTVSPWY peptide.J Mater Sci Mater Med. 2017 Aug 31;28(10):152. doi: 10.1007/s10856-017-5963-y. J Mater Sci Mater Med. 2017. PMID: 28861765
-
Enhanced Bioactivity of α-Tocopheryl Succinate Based Block Copolymer Nanoparticles by Reduced Hydrophobicity.Macromol Biosci. 2016 Dec;16(12):1824-1837. doi: 10.1002/mabi.201600259. Epub 2016 Oct 14. Macromol Biosci. 2016. PMID: 27739627 Free PMC article.
References
-
- Accelrys. InsightII. Accelrys Inc; San Diego: 2001.
-
- Adam-Vizi V, Chinopoulos C. Bioenergetics and the formation of mitochondrial reactive oxygen species. Trends Pharmacol Sci. 2006;27:639–645. - PubMed
-
- Allen FH. The Cambridge Structural Database: a quarter of a million crystal structures and rising. Acta Crystallogr B. 2002;5:380–388. - PubMed
-
- Allen RG, Balin AK. Effects of oxygen on the antioxidant responses of normal and transformed cells. Exp Cell Res. 2003;289:307–316. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

