A novel Staphylococcus aureus biofilm phenotype mediated by the fibronectin-binding proteins, FnBPA and FnBPB
- PMID: 18375547
- PMCID: PMC2395027
- DOI: 10.1128/JB.00167-08
A novel Staphylococcus aureus biofilm phenotype mediated by the fibronectin-binding proteins, FnBPA and FnBPB
Abstract
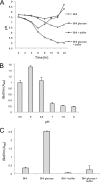
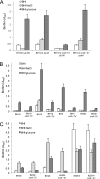
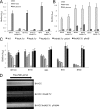
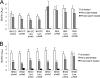
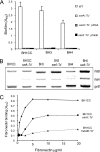
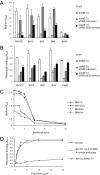
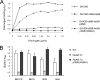
Device-associated infections involving biofilm remain a persistent clinical problem. We recently reported that four methicillin-resistant Staphylococcus aureus (MRSA) strains formed biofilm independently of the icaADBC-encoded exopolysaccharide. Here, we report that MRSA biofilm development was promoted under mildly acidic growth conditions triggered by the addition of glucose to the growth medium. Loss of sortase, which anchors LPXTG-containing proteins to peptidoglycan, reduced the MRSA biofilm phenotype. Furthermore introduction of mutations in fnbA and fnbB, which encode the LPXTG-anchored multifunctional fibrinogen and fibronectin-binding proteins, FnBPA and FnBPB, reduced biofilm formation by several MRSA strains. However, these mutations had no effect on biofilm formation by methicillin-sensitive S. aureus strains. FnBP-promoted biofilm occurred at the level of intercellular accumulation and not primary attachment. Mutation of fnbA or fnbB alone did not substantially affect biofilm, and expression of either gene alone from a complementing plasmid in fnbA fnbB mutants restored biofilm formation. FnBP-promoted biofilm was dependent on the integrity of SarA but not through effects on fnbA or fnbB transcription. Using plasmid constructs lacking regions of FnBPA to complement an fnbAB mutant revealed that the A domain alone and not the domain required for fibronectin binding could promote biofilm. Additionally, an A-domain N304A substitution that abolished fibrinogen binding did not affect biofilm. These data identify a novel S. aureus biofilm phenotype promoted by FnBPA and FnBPB which is apparently independent of the known ligand-binding activities of these multifunctional surface proteins.
Figures


Similar articles
-
Carriage of both the fnbA and fnbB genes and growth at 37 degrees C promote FnBP-mediated biofilm development in meticillin-resistant Staphylococcus aureus clinical isolates.J Med Microbiol. 2009 Apr;58(Pt 4):399-402. doi: 10.1099/jmm.0.005504-0. J Med Microbiol. 2009. PMID: 19273632
-
Fibronectin-binding proteins are required for biofilm formation by community-associated methicillin-resistant Staphylococcus aureus strain LAC.FEMS Microbiol Lett. 2014 Apr;353(2):157-64. doi: 10.1111/1574-6968.12424. Epub 2014 Apr 10. FEMS Microbiol Lett. 2014. PMID: 24628034
-
Subdomains N2N3 of fibronectin binding protein A mediate Staphylococcus aureus biofilm formation and adherence to fibrinogen using distinct mechanisms.J Bacteriol. 2013 Jun;195(11):2675-83. doi: 10.1128/JB.02128-12. Epub 2013 Apr 5. J Bacteriol. 2013. PMID: 23564165 Free PMC article.
-
Methicillin resistance and the biofilm phenotype in Staphylococcus aureus.Front Cell Infect Microbiol. 2015 Jan 28;5:1. doi: 10.3389/fcimb.2015.00001. eCollection 2015. Front Cell Infect Microbiol. 2015. PMID: 25674541 Free PMC article. Review.
-
The remarkably multifunctional fibronectin binding proteins of Staphylococcus aureus.Eur J Clin Microbiol Infect Dis. 2016 Dec;35(12):1923-1931. doi: 10.1007/s10096-016-2763-0. Epub 2016 Sep 7. Eur J Clin Microbiol Infect Dis. 2016. PMID: 27604831 Review.
Cited by
-
Alternative modes of biofilm formation by plant-associated Bacillus cereus.Microbiologyopen. 2015 Jun;4(3):452-64. doi: 10.1002/mbo3.251. Epub 2015 Mar 31. Microbiologyopen. 2015. PMID: 25828975 Free PMC article.
-
Biofilm density and detection of biofilm-producing genes in methicillin-resistant Staphylococcus aureus strains.Folia Microbiol (Praha). 2013 Jan;58(1):47-52. doi: 10.1007/s12223-012-0175-9. Epub 2012 Jun 19. Folia Microbiol (Praha). 2013. PMID: 22711180
-
Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management.Clin Microbiol Rev. 2015 Jul;28(3):603-61. doi: 10.1128/CMR.00134-14. Clin Microbiol Rev. 2015. PMID: 26016486 Free PMC article. Review.
-
Enzyme-Functionalized Mesoporous Silica Nanoparticles to Target Staphylococcus aureus and Disperse Biofilms.Int J Nanomedicine. 2021 Mar 8;16:1929-1942. doi: 10.2147/IJN.S293190. eCollection 2021. Int J Nanomedicine. 2021. PMID: 33727807 Free PMC article.
-
The sortase A substrates FnbpA, FnbpB, ClfA and ClfB antagonize colony spreading of Staphylococcus aureus.PLoS One. 2012;7(9):e44646. doi: 10.1371/journal.pone.0044646. Epub 2012 Sep 7. PLoS One. 2012. PMID: 22970276 Free PMC article.
References
-
- Bruckner, R. 1997. Gene replacement in Staphylococcus carnosus and Staphylococcus xylosus. FEMS Microbiol. Lett. 1511-8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

