Pore-opening mechanism of the nicotinic acetylcholine receptor evinced by proton transfer
- PMID: 18376414
- PMCID: PMC2596065
- DOI: 10.1038/nsmb.1407
Pore-opening mechanism of the nicotinic acetylcholine receptor evinced by proton transfer
Abstract
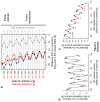
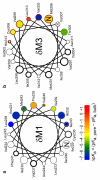
The conformational changes underlying cysteine-loop receptor channel gating remain elusive and controversial. We previously developed a single-channel electrophysiological method that allows structural inferences about the transient open-channel conformation to be made from the effect and properties of introduced charges on systematically engineered ionizable amino acids. Here we have applied this methodology to the entire M1 and M3 segments of the muscle nicotinic acetylcholine receptor, two transmembrane alpha-helices that pack against the pore-lining M2 alpha-helix. Together with our previous results on M2, these data suggest that the pore dilation that underlies channel opening involves only a subtle rearrangement of these three transmembrane helices. Such a limited conformational change seems optimal to allow rapid closed-open interconversion rates, and hence a fast postsynaptic response upon neurotransmitter binding. Thus, this receptor-channel seems to have evolved to take full advantage of the steep dependence of ion- and water-conduction rates on pore diameter that is characteristic of model hydrophobic nanopores.
Figures





References
-
- Unwin N. Refined structure of the nicotinic acetylcholine receptor at 4 Å resolution. J. Mol. Biol. 2005;346:967–989. - PubMed
-
- Dilger JP, Liu Y. Desensitization of acetylcholine receptors in BC3H-1 cells. Pflügers Arch. 1992;420:479–485. - PubMed
-
- Unwin N. Acetylcholine receptor channel imaged in the open state. Nature. 1995;373:37–43. - PubMed
-
- Arévalo E, Chiara DC, Forman SA, Cohen JB, Miller KW. Gating-enhanced accessibility of hydrophobic sites within the transmembrane region of the nicotinic acetylcholine receptor's δ-subunit. A time-resolved photolabeling study. J. Biol. Chem. 2005;280:13631–13640. - PubMed
-
- Sansom MSP. Twist to open. Current Biology. 1995;5:373–375. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

