Role of NADPH oxidase and Stat3 in statin-mediated protection against diabetic retinopathy
- PMID: 18378570
- PMCID: PMC2819293
- DOI: 10.1167/iovs.08-1754
Role of NADPH oxidase and Stat3 in statin-mediated protection against diabetic retinopathy
Abstract
Purpose: Inhibitors of 3-hydroxy-3-methylglutaryl CoA reductase (statins) reduce signs of diabetic retinopathy in diabetic patients and animals. Indirect clinical evidence supports the actions of statins in improving cardiovascular function, but the mechanisms of their protective actions in the retina are not understood. Prior studies have implicated oxidative stress and NADPH oxidase-mediated activation of signal transducer and activator of transcription 3 (STAT3) in diabetes-induced increases in expression of vascular endothelial growth factor (VEGF) and intercellular adhesion molecule (ICAM)-1 and breakdown of the blood-retinal barrier (BRB). Because statins are known to be potent antioxidants, the hypothesis for the current study was that the protective effects of statins in preventing diabetic retinopathy involve blockade of diabetes-induced activation of NADPH oxidase and STAT3.
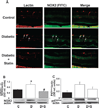
Methods: The hypothesis was tested by experiments in which rats with streptozotocin (STZ)-induced diabetes and retinal endothelial cells maintained in high-glucose medium were treated with simvastatin. Blood-retinal barrier (BRB) function was assayed by determining extravasation of albumin. Oxidative stress was assayed by measuring lipid peroxidation, protein nitration of tyrosine, dihydroethidine oxidation, and chemiluminescence. Immunoprobe techniques were used to determine the levels of NADPH oxidase subunit expression and STAT3 activation.
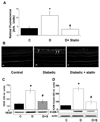
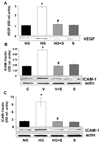
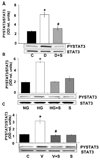
Results: These studies showed that simvastatin blocks diabetes or high-glucose-induced increases in VEGF and ICAM-1 and preserves the BRB by a process involving blockade of diabetes/high-glucose-induced activation of STAT3 and NADPH oxidase. Statin treatment also prevents diabetes-induced increases in expression of the NADPH oxidase catalytic and subunit NOX2.
Conclusions: These results suggest that simvastatin protects against the early signs of diabetic retinopathy by preventing NADPH oxidase-mediated activation of STAT3.
Figures
References
-
- Antonetti DA, Lieth E, Barber AJ, Gardner TW. Molecular mechanisms of vascular permeability in diabetic retinopathy. Semin Ophthalmol. 1999;14:240–248. - PubMed
-
- Caldwell RB, Bartoli M, Behzadian MA, et al. Vascular endothelial growth factor and diabetic retinopathy: role of oxidative stress. Curr Drug Targets. 2005;6:511–524. - PubMed
-
- Obrosova IG, Minchenko AG, Marinescu V, et al. Antioxidants attenuate early up regulation of retinal vascular endothelial growth factor in streptozotocin-diabetic rats. Diabetologia. 2001;44:1102–1110. - PubMed
-
- El-Remessy AB, Behzadian MA, Abou-Mohamed G, Franklin T, Cald-well RW, Caldwell RB. Experimental diabetes causes breakdown of the blood-retina barrier by a mechanism involving tyrosine nitration and increases in expression of vascular endothelial growth factor and urokinase plasminogen activator receptor. Am J Pathol. 2003;162:1995–2004. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

