Persistent inward currents in spinal motoneurons and their influence on human motoneuron firing patterns
- PMID: 18381974
- PMCID: PMC3326417
- DOI: 10.1177/1073858408314986
Persistent inward currents in spinal motoneurons and their influence on human motoneuron firing patterns
Abstract
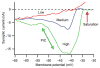
Persistent inward currents (PICs) are present in many types of neurons and likely have diverse functions. In spinal motoneurons, PICs are especially strong, primarily located in dendritic regions, and subject to particularly strong neuromodulation by the monoamines serotonin and norepinephrine. Because motoneurons drive muscle fibers, it has been possible to study the functional role of their PICs in motor output and to identify PIC-mediated effects on motoneuron firing patterns in human subjects. The PIC markedly amplifies synaptic input, up to fivefold or more, depending on the level of monoaminergic input. PICs also tend to greatly prolong input time course, allowing brief inputs to initiate long-lasting self-sustained firing (i.e., bistable behavior). PIC deactivation usually requires inhibitory input and PIC amplitude can increase to repeated activation. All of these behaviors markedly increase motoneuron excitability. Thus, in the absence of monoaminergic input, motoneuron excitability is very low. Yet PICs have another effect: once active, they tend to sharply limit efficacy of additional synaptic input. All of these PIC effects have been detected in motoneuron firing patterns in human subjects and, hence, PICs are likely a fundamental component of normal motor output.
Figures

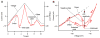



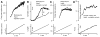

References
-
- Alaburda A, Perrier JF, Hounsgaard J. Mechanisms causing plateau potentials in spinal motoneurones. Adv Exp Med Biol. 2002;508:219–26. - PubMed
-
- Alvarez FJ, Pearson JC, Harrington D, Dewey D, Torbeck L, Fyffe RE. Distribution of 5-hydroxytryptamine-immunoreactive boutons on alpha-motoneurons in the lumbar spinal cord of adult cats. J Comp Neurol. 1998;393:69–83. - PubMed
-
- Aston-Jones G, Chen S, Zhu Y, Oshinsky ML. A neural circuit for circadian regulation of arousal. Nat Neurosci. 2001;4:732–8. - PubMed
-
- Ballou EW, Smith WB, Anelli R, Heckman CJ. Measuring dendritic distribution of membrane proteins. J Neurosci Methods. 2006;156:257–66. - PubMed
-
- Bennett DJ, Hultborn H, Fedirchuk B, Gorassini M. Short-term plasticity in hindlimb motoneurons of decerebrate cats. J Neurophysiol. 1998a;80:2038–45. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

