The desmosome and pemphigus
- PMID: 18386043
- PMCID: PMC2413110
- DOI: 10.1007/s00418-008-0420-0
The desmosome and pemphigus
Abstract
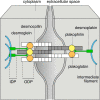
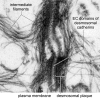
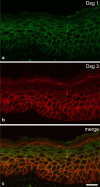
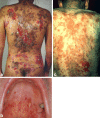
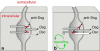
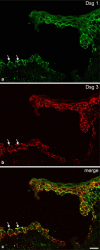
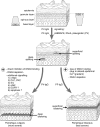
Desmosomes are patch-like intercellular adhering junctions ("maculae adherentes"), which, in concert with the related adherens junctions, provide the mechanical strength to intercellular adhesion. Therefore, it is not surprising that desmosomes are abundant in tissues subjected to significant mechanical stress such as stratified epithelia and myocardium. Desmosomal adhesion is based on the Ca(2+)-dependent, homo- and heterophilic transinteraction of cadherin-type adhesion molecules. Desmosomal cadherins are anchored to the intermediate filament cytoskeleton by adaptor proteins of the armadillo and plakin families. Desmosomes are dynamic structures subjected to regulation and are therefore targets of signalling pathways, which control their molecular composition and adhesive properties. Moreover, evidence is emerging that desmosomal components themselves take part in outside-in signalling under physiologic and pathologic conditions. Disturbed desmosomal adhesion contributes to the pathogenesis of a number of diseases such as pemphigus, which is caused by autoantibodies against desmosomal cadherins. Beside pemphigus, desmosome-associated diseases are caused by other mechanisms such as genetic defects or bacterial toxins. Because most of these diseases affect the skin, desmosomes are interesting not only for cell biologists who are inspired by their complex structure and molecular composition, but also for clinical physicians who are confronted with patients suffering from severe blistering skin diseases such as pemphigus. To develop disease-specific therapeutic approaches, more insights into the molecular composition and regulation of desmosomes are required.
Figures

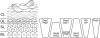


References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PMC', 'value': 'PMC41522', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC41522/'}, {'type': 'PubMed', 'value': '7604000', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7604000/'}]}
- Aberle H, Bierkamp C, Torchard D, Serova O, Wagner T, Natt E, Wirsching J, Heidkamper C, Montagna M, Lynch HT et al (1995) The human plakoglobin gene localizes on chromosome 17q21 and is subjected to loss of heterozygosity in breast and ovarian cancers. Proc Natl Acad Sci USA 92:6384–6388 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '17065638', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/17065638/'}]}
- Ahmed AR, Spigelman Z, Cavacini LA, Posner MR (2006) Treatment of pemphigus vulgaris with rituximab and intravenous immune globulin. New Engl J Med 355:1772–1779 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '17965884', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/17965884/'}]}
- Akat K, Bleck CK, Lee YM, Haselmann-Weiss U, Kartenbeck J (2008) Characterization of a novel type of adherens junction in meningiomas and the derived cell line HBL-52. Cell Tissue Res 331:401–412 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PMC', 'value': 'PMC32271', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC32271/'}, {'type': 'PubMed', 'value': '11294891', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11294891/'}]}
- Akhtar N, Hotchin NA (2001) RAC1 regulates adherens junctions through endocytosis of E-cadherin. Molec Biol Cell 12:847–862 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '18064004', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/18064004/'}]}
- Al-Amoudi A, Diez DC, Betts MJ, Frangakis AS (2007) The molecular architecture of cadherins in native epidermal desmosomes. Nature 450:832–837 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

