Development of the movement of the epiglottis in infant and juvenile pigs
- PMID: 18387794
- PMCID: PMC2574016
- DOI: 10.1016/j.zool.2007.10.002
Development of the movement of the epiglottis in infant and juvenile pigs
Abstract
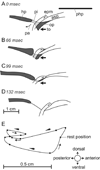
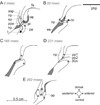
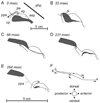
Although backward folding of the epiglottis is one of the signal events of the mammalian adult swallow, the epiglottis does not fold during the infant swallow. How this functional change occurs is unknown, but we hypothesize that a change in swallow mechanism occurs with maturation, prior to weaning. Using videofluoroscopy, we found three characteristic patterns of swallowing movement at different ages in the pig: an infant swallow, a transitional swallow and a post-weaning (juvenile or adult) swallow. In animals of all ages, the dorsal region of the epiglottis and larynx was held in an intranarial position by a muscular sphincter formed by the palatopharyngeal arch. In the infant swallow, increasing pressure in the oropharynx forced a liquid bolus through the piriform recesses on either side of a relatively stationary epiglottis into the esophagus. As the infant matured, the palatopharyngeal arch and the soft palate elevated at the beginning of the swallow, so exposing a larger area of the epiglottis to bolus pressure. In transitional swallows, the epiglottis was tilted backward relatively slowly by a combination of bolus pressure and squeezing of the epiglottis by closure of the palatopharyngeal sphincter. The bolus, however, traveled alongside but never over the tip of the epiglottis. In the juvenile swallow, the bolus always passed over the tip of the epiglottis. The tilting of the epiglottis resulted from several factors, including the action of the palatopharyngeal sphincter, higher bolus pressure exerted on the epiglottis and the allometry of increased size. In both transitional and juvenile swallows, the subsequent relaxation of the palatopharyngeal sphincter released the epiglottis, which sprang back to its original intranarial position.
Figures





References
-
- Amis TC, O'Neill N, Van der Touw T, Brancatisano A. Electromyographic activity of the hyoepiglotticus muscle in dogs. Respir. Physiol. 1996;104:159–167. - PubMed
-
- Amri M, Lamkadem M, Car A. Activity of extrinsic tongue muscles during swallowing in sheep. Brain Research. 1989;503:141–143. - PubMed
-
- Ardran GM, Kemp FH. The protection of the laryngeal airway during swallowing. Br. J. Radiol. 1952;25:406–416. - PubMed
-
- Ardran GM, Kemp FH, Ride W. A radiographic analysis of mastication and swallowing in the domestic rabbit: Oryctolagus cuniculus. Proc. Zool. Soc. London. 1957;130:257–274.
-
- Ardran GM, Kemp FH, Lind J. A cineradiographic study of bottle feeding. Br. J. Radiol. 1958;31:11–22. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

