The class V myosin motor protein, Myo2, plays a major role in mitochondrial motility in Saccharomyces cerevisiae
- PMID: 18391073
- PMCID: PMC2287292
- DOI: 10.1083/jcb.200709099
The class V myosin motor protein, Myo2, plays a major role in mitochondrial motility in Saccharomyces cerevisiae
Abstract
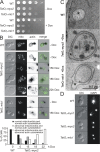
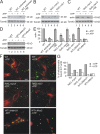
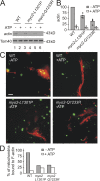
The actin cytoskeleton is essential for polarized, bud-directed movement of cellular membranes in Saccharomyces cerevisiae and thus ensures accurate inheritance of organelles during cell division. Also, mitochondrial distribution and inheritance depend on the actin cytoskeleton, though the precise molecular mechanisms are unknown. Here, we establish the class V myosin motor protein, Myo2, as an important mediator of mitochondrial motility in budding yeast. We found that mutants with abnormal expression levels of Myo2 or its associated light chain, Mlc1, exhibit aberrant mitochondrial morphology and loss of mitochondrial DNA. Specific mutations in the globular tail of Myo2 lead to aggregation of mitochondria in the mother cell. Isolated mitochondria lacking functional Myo2 are severely impaired in their capacity to bind to actin filaments in vitro. Time-resolved fluorescence microscopy revealed a block of bud-directed anterograde mitochondrial movement in cargo binding-defective myo2 mutant cells. We conclude that Myo2 plays an important and direct role for mitochondrial motility and inheritance in budding yeast.
Figures


Comment in
-
Pushing for answers: is myosin V directly involved in moving mitochondria?J Cell Biol. 2008 Apr 7;181(1):15-8. doi: 10.1083/jcb.200803064. J Cell Biol. 2008. PMID: 18391069 Free PMC article.
References
-
- Altmann, K., M. Dürr, and B. Westermann. 2007. Saccharomyces cerevisiae as a model organism to study mitochondrial biology: general considerations and basic procedures. Methods Mol. Biol. 372:81–90. - PubMed
-
- Bereiter-Hahn, J. 1990. Behavior of mitochondria in the living cell. Int. Rev. Cytol. 122:1–63. - PubMed
-
- Berger, K.H., and M.P. Yaffe. 2000. Mitochondrial DNA inheritance in Saccharomyces cerevisiae. Trends Microbiol. 8:508–513. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

