Mechanical homeostasis is altered in uterine leiomyoma
- PMID: 18395046
- PMCID: PMC2696475
- DOI: 10.1016/j.ajog.2007.11.057
Mechanical homeostasis is altered in uterine leiomyoma
Abstract
Objective: Uterine leiomyoma produce an extracellular matrix (ECM) that is abnormal in its volume, content, and structure. Alterations in ECM can modify mechanical stress on cells and lead to activation of Rho-dependent signaling and cell growth. Here we sought to determine whether the altered ECM that is produced by leiomyoma was accompanied by an altered state of mechanical homeostasis.
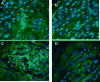
Study design: We measured the mechanical response of paired leiomyoma and myometrial samples and performed immunogold, confocal microscopy, and immunohistochemical analyses.
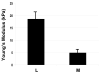
Results: Leiomyoma were significantly stiffer than matched myometrium. The increased stiffness was accompanied by alteration of the ECM, cell shape, and cytoskeleton in leiomyoma, compared with myometrial samples from the same uterus. Levels of AKAP13, a protein that is known to activate Rho, were increased in leiomyoma compared to myometrium. AKAP13 was associated with cytoskeletal filaments of immortalized leiomyoma cells.
Conclusion: Leiomyoma cells are exposed to increased mechanical loading and show structural and biochemical features that are consistent with the activation of solid-state signaling.
Figures










References
-
- Huang S, Ingber DE. Cell tension, matrix mechanics, and cancer development. Cancer Cell. 2005;8:175–76. - PubMed
-
- Alenghat FJ, Ingber DE. Mechanotransduction: all signals point to cytoskeleton, matrix and integrins. Sci STKE. 2002;119:Pe6. - PubMed
-
- Geiger B, Bershadsky A. Exploring the neighborhood: adhesion-coupled cell mechanosensors. Cell. 2002;110:139–142. - PubMed
-
- Kawamura S, Miyamoto S, Brown JH. Initiation and transduction of stretch-induced RhoA and Rac1 activation through caveolae. J Biol Chem. 2003;278:31111–7. - PubMed
-
- Ruwhof C, van der Laarse A. Mechanical stress-induced cardiac hypertrophy: mechanisms and signal transduction pathways. Cardiovascular Res. 2000;47:223–37. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

