Cathepsin D--many functions of one aspartic protease
- PMID: 18396408
- PMCID: PMC2635020
- DOI: 10.1016/j.critrevonc.2008.02.008
Cathepsin D--many functions of one aspartic protease
Abstract
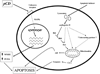
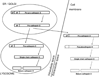
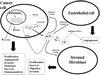
For years, it has been held that cathepsin D (CD) is involved in rather non-specific protein degradation in a strongly acidic milieu of lysosomes. Studies with CD knock-out mice revealed that CD is not necessary for embryonal development, but it is indispensable for postnatal tissue homeostasis. Mutation that abolishes CD enzymatic activity causes neuronal ceroid lipofuscinosis (NCL) characterized by severe neurodegeneration, developmental regression, visual loss and epilepsy in both animals and humans. In the last decade, however, an increasing number of studies demonstrated that enzymatic function of CD is not restricted solely to acidic milieu of lysosomes with important consequences in regulation of apoptosis. In addition to CD enzymatic activity, it has been shown that apoptosis is also regulated by catalytically inactive mutants of CD which suggests that CD interacts with other important molecules and influences cell signaling. Moreover, procathepsin D (pCD), secreted from cancer cells, acts as a mitogen on both cancer and stromal cells and stimulates their pro-invasive and pro-metastatic properties. Numerous studies found that pCD/CD level represents an independent prognostic factor in a variety of cancers and is therefore considered to be a potential target of anti-cancer therapy. Studies dealing with functions of cathepsin D are complicated by the fact that there are several simultaneous forms of CD in a cell-pCD, intermediate enzymatically active CD and mature heavy and light chain CD. It became evident that these forms may differently regulate the above-mentioned processes. In this article, we review the possible functions of CD and its various forms in cells and organisms during physiological and pathological conditions.
Figures
References
-
- Hasilik A, Neufeld EF. Biosynthesis of lysosomal enzymes in fibroblasts. Synthesis as precursors of higher molecular weight. J Biol Chem. 1980;255:4937–4945. - PubMed
-
- Kornfeld S. Lysosomal enzyme targeting. Biochem Soc Trans. 1990;18:367–374. - PubMed
-
- Hasilik A, Neufeld EF. Biosynthesis of lysosomal enzymes in fibroblasts. Phosphorylation of mannose residues. J Biol Chem. 1980;255:4946–4950. - PubMed
-
- Fortenberry SC, Schorey JS, Chirgwin JM. Role of glycosylation in the expression of human procathepsin D. J Cell Sci. 1995;108:2001–2006. - PubMed
-
- von Figura K, Hasilik A. Lysosomal enzymes and their receptors. Annu Rev Biochem. 1986;55:167–193. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

