RGT, a synthetic peptide corresponding to the integrin beta 3 cytoplasmic C-terminal sequence, selectively inhibits outside-in signaling in human platelets by disrupting the interaction of integrin alpha IIb beta 3 with Src kinase
- PMID: 18398066
- PMCID: PMC2481538
- DOI: 10.1182/blood-2007-09-110437
RGT, a synthetic peptide corresponding to the integrin beta 3 cytoplasmic C-terminal sequence, selectively inhibits outside-in signaling in human platelets by disrupting the interaction of integrin alpha IIb beta 3 with Src kinase
Abstract
Mutational analysis has established that the cytoplasmic tail of the integrin beta 3 subunit binds c-Src (termed as Src in this study) and is critical for bidirectional integrin signaling. Here we show in washed human platelets that a cell-permeable, myristoylated RGT peptide (myr-RGT) corresponding to the integrin beta 3 C-terminal sequence dose-dependently inhibited stable platelet adhesion and spreading on immobilized fibrinogen, and fibrin clot retraction as well. Myr-RGT also inhibited the aggregation-dependent platelet secretion and secretion-dependent second wave of platelet aggregation induced by adenosine diphosphate, ristocetin, or thrombin. Thus, myr-RGT inhibited integrin outside-in signaling. In contrast, myr-RGT had no inhibitory effect on adenosine diphosphate-induced soluble fibrinogen binding to platelets that is dependent on integrin inside-out signaling. Furthermore, the RGT peptide induced dissociation of Src from integrin beta 3 and dose-dependently inhibited the purified recombinant beta 3 cytoplasmic domain binding to Src-SH3. In addition, phosphorylation of the beta 3 cytoplasmic tyrosines, Y(747) and Y(759), was inhibited by myr-RGT. These data indicate an important role for beta 3-Src interaction in outside-in signaling. Thus, in intact human platelets, disruption of the association of Src with beta 3 and selective blockade of integrin alpha IIb beta 3 outside-in signaling by myr-RGT suggest a potential new antithrombotic strategy.
Figures
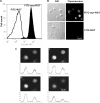
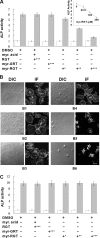
 ) was quantified by a PNPP assay. Data are arranged as in panel A.
) was quantified by a PNPP assay. Data are arranged as in panel A.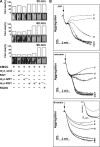

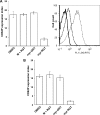


Comment in
-
Outside-in: peptide versus integrin.Blood. 2008 Aug 1;112(3):453-4. doi: 10.1182/blood-2008-05-153585. Blood. 2008. PMID: 18650453 No abstract available.
References
-
- Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673–687. - PubMed
-
- Ginsberg MH, Partridge A, Shattil SJ. Integrin regulation. Curr Opin Cell Biol. 2005;17:509–516. - PubMed
-
- Tadokoro S, Shattil SJ, Eto K, et al. Talin binding to integrin β tails: a final common step in integrin activation. Science. 2003;302:103–106. - PubMed
-
- Vinogradova O, Velyvis A, Velyviene A, et al. A structural mechanism of integrin αIIbβ3 “in-side-out” activation as regulated by its cytoplasmic face. Cell. 2002;110:587–597. - PubMed
-
- Shattil SJ, Kashiwagi H, Pampori N. Integrin signaling: the platelet paradigm. Blood. 1998;91:2645–2657. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

