Evaluating human papillomavirus vaccination programs in Canada: should provincial healthcare pay for voluntary adult vaccination?
- PMID: 18402671
- PMCID: PMC2373297
- DOI: 10.1186/1471-2458-8-114
Evaluating human papillomavirus vaccination programs in Canada: should provincial healthcare pay for voluntary adult vaccination?
Abstract
Background: Recently, provincial health programs in Canada and elsewhere have begun rolling out vaccination against human papillomavirus for girls aged 9-13. While vaccination is voluntary, the cost of vaccination is waived, to encourage parents to have their daughters vaccinated. Adult women who are eligible for the vaccine may still receive it, but at a cost of approximately CAN$400. Given the high efficacy and immunogenicity of the vaccine, the possibility of eradicating targeted types of the virus may be feasible, assuming the vaccination programs are undertaken strategically.
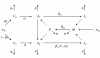
Methods: We develop a mathematical model to describe the epidemiology of vaccination against human papillomavirus, accounting for a widespread childhood vaccination program that may be supplemented by voluntary adult vaccination. A stability analysis is performed to determine the stability of the disease-free equilibrium. The critical vaccine efficacy and immunogenicity thresholds are derived, and the minimum level of adult vaccination required for eradication of targeted types is determined.
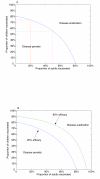
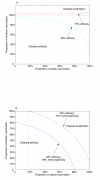
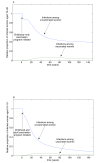
Results: We demonstrate that eradication of targeted types is indeed feasible, although the burden of coverage for a childhood-only vaccination program may be high. However, if a small, but non-negligible, proportion of eligible adults can be vaccinated, then the possibility of eradication of targeted types becomes much more favourable. We provide a threshold for eradication in general communities and illustrate the results with numerical simulations. We also investigate the effects of suboptimal efficacy and immunogenicity and show that there is a critical efficacy below which eradication of targeted types is not possible. If eradication is possible, then there is a critical immunogenicity such that even 100% childhood vaccination will not eradicate the targeted types of the virus and must be supplemented with voluntary adult vaccination. However, the level of adult vaccination coverage required is modest and may be achieved simply by removing the cost burden to vaccination.
Conclusion: We recommend that provincial healthcare programs should pay for voluntary adult vaccination for women aged 14-26. However, it should be noted that our model results are preliminary, in that we have made a number of simplifying assumptions, including a lack of age-dependency in sexual partner rates, a lack of sexual activity outside of the vaccine age-range among females and a uniform age of sexual debut; thus, further work is desired to enhance the external generalisability of our results.
Figures
References
-
- eMedicine, Human papillomavirus http://www.emedicine.com/med/topic1037.htm
-
- Koutsky LA, Galloway DA, Holmes KK. Epidemiology of genital human papillomavirus infection. Epidemiol Rev. 1988;10:122–163. - PubMed
-
- Richardson H, Kelsall G, Tellier P, Voyer H, Abrahamowicz M, Ferenczy A, Coutlée F, Franco EL. The Natural History of Type-specific Human Papillomavirus Infections in Female University Students. Cancer Epidemiology, Biomarkers & Prevention. 2003;12:485–490. - PubMed
-
- Ontario Ministry of Health and Long-Term Care, Ontario's Grade 8 Vaccination program http://www.health.gov.on.ca/hpv
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

