Identification and characterization of a novel folliculin-interacting protein FNIP2
- PMID: 18403135
- PMCID: PMC2727720
- DOI: 10.1016/j.gene.2008.02.022
Identification and characterization of a novel folliculin-interacting protein FNIP2
Abstract
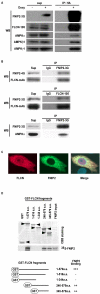
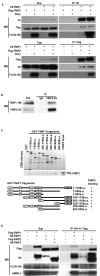
Birt-Hogg-Dube' syndrome characterized by increased risk for renal neoplasia is caused by germline mutations in the BHD/FLCN gene encoding a novel tumor suppressor protein, folliculin(FLCN), which interacts with FNIP1 and 5'-AMP-activated protein kinase(AMPK). Here we report the identification and characterization of a novel FNIP1 homolog FNIP2 that also interacts with FLCN and AMPK. C-terminally-deleted FLCN mutants, similar to those produced by naturally-occurring germline mutations in BHD patients, were unable to bind FNIP2. These data taken together with our previous results that demonstrated FNIP1 binding to the C-terminus of FLCN suggest that FLCN tumor suppressor function may be facilitated by interactions with both FNIP1 and FNIP2 through its C-terminus. Furthermore, we demonstrate that FNIP1 and FNIP2 are able to form homo- or heteromeric multimers suggesting that they may function independently or cooperatively with FLCN. Differential expression of FNIP1 and FNIP2 transcripts in some normal tissues may indicate tissue specificity for these homologs. Interestingly FNIP1 and FNIP2 were oppositely expressed in human clear cell renal cell carcinoma (RCC), and coordinately expressed in chromophobe RCC and oncocytoma, suggesting their differential function in different histologic variants of RCC.
Figures
Similar articles
-
Interaction of folliculin (Birt-Hogg-Dubé gene product) with a novel Fnip1-like (FnipL/Fnip2) protein.Oncogene. 2008 Sep 11;27(40):5339-47. doi: 10.1038/onc.2008.261. Epub 2008 Jul 28. Oncogene. 2008. PMID: 18663353
-
Folliculin-interacting proteins Fnip1 and Fnip2 play critical roles in kidney tumor suppression in cooperation with Flcn.Proc Natl Acad Sci U S A. 2015 Mar 31;112(13):E1624-31. doi: 10.1073/pnas.1419502112. Epub 2015 Mar 16. Proc Natl Acad Sci U S A. 2015. PMID: 25775561 Free PMC article.
-
Birt-Hogg-Dubé syndrome: Clinical and molecular aspects of recently identified kidney cancer syndrome.Int J Urol. 2016 Mar;23(3):204-10. doi: 10.1111/iju.13015. Epub 2015 Nov 25. Int J Urol. 2016. PMID: 26608100 Review.
-
Folliculin encoded by the BHD gene interacts with a binding protein, FNIP1, and AMPK, and is involved in AMPK and mTOR signaling.Proc Natl Acad Sci U S A. 2006 Oct 17;103(42):15552-7. doi: 10.1073/pnas.0603781103. Epub 2006 Oct 6. Proc Natl Acad Sci U S A. 2006. PMID: 17028174 Free PMC article.
-
FLCN: The causative gene for Birt-Hogg-Dubé syndrome.Gene. 2018 Jan 15;640:28-42. doi: 10.1016/j.gene.2017.09.044. Epub 2017 Sep 29. Gene. 2018. PMID: 28970150 Free PMC article. Review.
Cited by
-
Folliculin interacts with p0071 (plakophilin-4) and deficiency is associated with disordered RhoA signalling, epithelial polarization and cytokinesis.Hum Mol Genet. 2012 Dec 15;21(24):5268-79. doi: 10.1093/hmg/dds378. Epub 2012 Sep 10. Hum Mol Genet. 2012. PMID: 22965878 Free PMC article.
-
Ciliary localization of folliculin mediated via a kinesin-2-binding motif is required for its functions in mTOR regulation and tumor suppression.FEBS Lett. 2021 Jan;595(1):123-132. doi: 10.1002/1873-3468.13959. Epub 2020 Nov 20. FEBS Lett. 2021. PMID: 33064845 Free PMC article.
-
Familial renal cancer: molecular genetics and surgical management.Int J Surg Oncol. 2011;2011:658767. doi: 10.1155/2011/658767. Epub 2011 Aug 22. Int J Surg Oncol. 2011. PMID: 22312516 Free PMC article.
-
Emerging Link between Tsc1 and FNIP Co-Chaperones of Hsp90 and Cancer.Biomolecules. 2022 Jul 1;12(7):928. doi: 10.3390/biom12070928. Biomolecules. 2022. PMID: 35883484 Free PMC article. Review.
-
Birt-Hogg-Dubé renal tumors are genetically distinct from other renal neoplasias and are associated with up-regulation of mitochondrial gene expression.BMC Med Genomics. 2010 Dec 16;3:59. doi: 10.1186/1755-8794-3-59. BMC Med Genomics. 2010. PMID: 21162720 Free PMC article.
References
-
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J. Mol. Biol. 1990;215:403–410. - PubMed
-
- Birt AR, Hogg GR, Dube WJ. Hereditary multiple fibrofolliculomas with trichodiscomas and acrochordons. Arch. Dermatol. 1977;113:1674–1677. - PubMed
-
- Inoki K, Corradetti MN, Guan KL. Dysregulation of the TSC-mTOR pathway in human disease. Nat. Genet. 2005;37:19–24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

