A novel mechanism for CCR4 in the regulation of macrophage activation in bleomycin-induced pulmonary fibrosis
- PMID: 18403600
- PMCID: PMC2329831
- DOI: 10.2353/ajpath.2008.070832
A novel mechanism for CCR4 in the regulation of macrophage activation in bleomycin-induced pulmonary fibrosis
Abstract
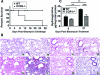
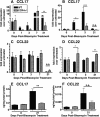
Macrophage polarization into M1 or M2 phenotypes dictates the nature, duration, and severity of an inflammatory response. The objective of this study was to examine the role of CC chemokine receptor 4 (CCR4) in macrophage polarization during pulmonary oxidative injury in wild-type [WT (CCR4(+/+))] and CCR4-deficient (CCR4(-/-)) mice. Intrapulmonary administration of bleomycin sulfate provoked lethal inflammatory and fibrotic responses in WT (CCR4(+/+)) mice, but such responses were absent in CCR4(-/-) mice. Transcript and protein analyses of alveolar and bone marrow-derived macrophages showed that cells isolated from CCR4(-/-) mice did not exhibit CCL17-dependent M1 activation in response to bleomycin. Instead, CCR4(-/-) macrophages showed an M2 phenotype characterized by significantly elevated expression of arginase 1 and FIZZ1 (found in inflammatory zone 1), particularly during the peak of pulmonary inflammation. Compared with WT (CCR4(+/+)) mice, CCR4(-/-) mice exhibited a significant increase in the expression of the nonsignaling CC chemokine scavenging receptor D6 in whole lung samples and isolated macrophages. Thus, these results demonstrate that CCL17-dependent activation of CCR4 in macrophages plays a central role in free radical-induced pulmonary injury and repair.
Figures


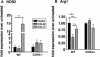




References
-
- King TE, Jr, Tooze JA, Schwarz MI, Brown KR, Cherniack RM. Predicting survival in idiopathic pulmonary fibrosis: scoring system and survival model. Am J Respir Crit Care Med. 2001;164:1171–1181. - PubMed
-
- Strieter RM, Keane MP, Burdick MD, Sakkour A, Murray LA, Belperio JA. The role of CXCR2/CXCR2 ligands in acute lung injury. Curr Drug Targets Inflamm Allergy. 2005;4:299–303. - PubMed
-
- Tajima S, Oshikawa K, Tominaga S, Sugiyama Y. The increase in serum soluble ST2 protein upon acute exacerbation of idiopathic pulmonary fibrosis. Chest. 2003;124:1206–1214. - PubMed
-
- Charo IF, Ransohoff RM. The many roles of chemokines and chemokine receptors in inflammation. N Engl J Med. 2006;354:610–621. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

