The role of ATP and adenosine in the brain under normoxic and ischemic conditions
- PMID: 18404443
- PMCID: PMC2072927
- DOI: 10.1007/s11302-007-9085-8
The role of ATP and adenosine in the brain under normoxic and ischemic conditions
Abstract
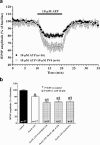
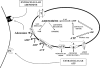
By taking advantage of some recently synthesized compounds that are able to block ecto-ATPase activity, we demonstrated that adenosine triphosphate (ATP) in the hippocampus exerts an inhibitory action independent of its degradation to adenosine. In addition, tonic activation of P2 receptors contributes to the normally recorded excitatory neurotransmission. The role of P2 receptors becomes critical during ischemia when extracellular ATP concentrations increase. Under such conditions, P2 antagonism is protective. Although ATP exerts a detrimental role under ischemia, it also exerts a trophic role in terms of cell division and differentiation. We recently reported that ATP is spontaneously released from human mesenchymal stem cells (hMSCs) in culture. Moreover, it decreases hMSC proliferation rate at early stages of culture. Increased hMSC differentiation could account for an ATP-induced decrease in cell proliferation. ATP as a homeostatic regulator might exert a different effect on cell trophism according to the rate of its efflux and receptor expression during the cell life cycle. During ischemia, adenosine formed by intracellular ATP escapes from cells through the equilibrative transporter. The protective role of adenosine A(1) receptors during ischemia is well accepted. However, the use of selective A(1) agonists is hampered by unwanted peripheral effects, thus attention has been focused on A(2A) and A(3) receptors. The protective effects of A(2A) antagonists in brain ischemia may be largely due to reduced glutamate outflow from neurones and glial cells. Reduced activation of p38 mitogen-activated protein kinases that are involved in neuronal death through transcriptional mechanisms may also contribute to protection by A(2A) antagonism. Evidence that A(3) receptor antagonism may be protective after ischemia is also reported.
Figures

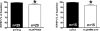
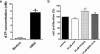
Similar articles
-
Purinergic signalling in brain ischemia.Neuropharmacology. 2016 May;104:105-30. doi: 10.1016/j.neuropharm.2015.11.007. Epub 2015 Nov 12. Neuropharmacology. 2016. PMID: 26581499 Review.
-
Purinergic modulation of glutamate release under ischemic-like conditions in the hippocampus.Neuroscience. 2007 Oct 12;149(1):99-111. doi: 10.1016/j.neuroscience.2007.07.035. Epub 2007 Aug 1. Neuroscience. 2007. PMID: 17850981
-
The Selective Antagonism of Adenosine A2B Receptors Reduces the Synaptic Failure and Neuronal Death Induced by Oxygen and Glucose Deprivation in Rat CA1 Hippocampus in Vitro.Front Pharmacol. 2018 Apr 24;9:399. doi: 10.3389/fphar.2018.00399. eCollection 2018. Front Pharmacol. 2018. PMID: 29740323 Free PMC article.
-
ATP-induced changes in rat skeletal muscle contractility.Int J Risk Saf Med. 2015;27 Suppl 1:S82-3. doi: 10.3233/JRS-150700. Int J Risk Saf Med. 2015. PMID: 26639725
-
Trophic effects of purines in neurons and glial cells.Prog Neurobiol. 1999 Dec;59(6):663-90. doi: 10.1016/s0301-0082(99)00017-9. Prog Neurobiol. 1999. PMID: 10845757 Review.
Cited by
-
The selective antagonism of P2X7 and P2Y1 receptors prevents synaptic failure and affects cell proliferation induced by oxygen and glucose deprivation in rat dentate gyrus.PLoS One. 2014 Dec 19;9(12):e115273. doi: 10.1371/journal.pone.0115273. eCollection 2014. PLoS One. 2014. PMID: 25526634 Free PMC article.
-
Role of the purinergic P2Y2 receptor in hippocampal function in mice.Eur Rev Med Pharmacol Sci. 2020 Nov;24(22):11858-11864. doi: 10.26355/eurrev_202011_23843. Eur Rev Med Pharmacol Sci. 2020. PMID: 33275273 Free PMC article.
-
Sawhorse waveform voltammetry for selective detection of adenosine, ATP, and hydrogen peroxide.Anal Chem. 2014 Aug 5;86(15):7486-93. doi: 10.1021/ac501229c. Epub 2014 Jul 15. Anal Chem. 2014. PMID: 25005825 Free PMC article.
-
Neuroprotection against hypoxia/ischemia: δ-opioid receptor-mediated cellular/molecular events.Cell Mol Life Sci. 2013 Jul;70(13):2291-303. doi: 10.1007/s00018-012-1167-2. Epub 2012 Sep 27. Cell Mol Life Sci. 2013. PMID: 23014992 Free PMC article. Review.
-
Vasoactive neuropeptides in clinical ophthalmology: An association with autoimmune retinopathy?Clin Ophthalmol. 2009;3:259-61. doi: 10.2147/opth.s5356. Epub 2009 Jun 2. Clin Ophthalmol. 2009. PMID: 19668576 Free PMC article.
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PMC', 'value': 'PMC1665813', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC1665813/'}, {'type': 'PubMed', 'value': '4339250', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/4339250/'}]}
- Burnstock G, Dumsday B, Smythe A (1972) Atropine resistant excitation of the urinary bladder: the possibility of transmission via nerves releasing a purine nucleotide. Br J Pharmacol 44:451–461 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '658131', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/658131/'}]}
- Burnstock G, Cocks T, Kasakov L et al (1978) Direct evidence for ATP release from non-adrenergic, non-cholinergic (“purinergic”) nerves in the guinea-pig taenia coli and bladder. Eur J Pharmacol 49:145–149 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PMC', 'value': 'PMC9389454', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC9389454/'}, {'type': 'PubMed', 'value': '11734617', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11734617/'}]}
- Fredholm BB, IJzerman AP, Jacobson KA et al (2001) International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol Rev 53:527–552 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '7724657', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7724657/'}]}
- Abbracchio MP, Burnstock G (1994) Purinoceptors: are there families of P2X and P2Y purinoceptors? Pharmacol Ther 64:445–475 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '12270951', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/12270951/'}]}
- North RA (2002) Molecular physiology of P2X receptors. Physiol Rev 82:1013–1067 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

