Sustained release of sphingosine 1-phosphate for therapeutic arteriogenesis and bone tissue engineering
- PMID: 18405965
- PMCID: PMC2711780
- DOI: 10.1016/j.biomaterials.2008.03.017
Sustained release of sphingosine 1-phosphate for therapeutic arteriogenesis and bone tissue engineering
Abstract
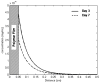
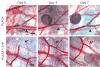
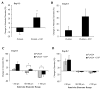
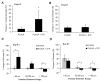
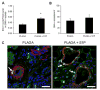
Sphingosine 1-phosphate (S1P) is a bioactive phospholipid that impacts migration, proliferation, and survival in diverse cell types, including endothelial cells, smooth muscle cells, and osteoblast-like cells. In this study, we investigated the effects of sustained release of S1P on microvascular remodeling and associated bone defect healing in vivo. The murine dorsal skinfold window chamber model was used to evaluate the structural remodeling response of the microvasculature. Our results demonstrated that 1:400 (w/w) loading and subsequent sustained release of S1P from poly(lactic-co-glycolic acid) (PLAGA) significantly enhanced lumenal diameter expansion of arterioles and venules after 3 and 7 days. Incorporation of 5-bromo-2-deoxyuridine (BrdU) at day 7 revealed significant increases in mural cell proliferation in response to S1P delivery. Additionally, three-dimensional (3D) scaffolds loaded with S1P (1:400) were implanted into critical-size rat calvarial defects, and healing of bony defects was assessed by radiograph X-ray, microcomputed tomography (muCT), and histology. Sustained release of S1P significantly increased the formation of new bone after 2 and 6 weeks of healing and histological results suggest increased numbers of blood vessels in the defect site. Taken together, these experiments support the use of S1P delivery for promoting microvessel diameter expansion and improving the healing outcomes of tissue-engineered therapies.
Figures
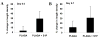


Similar articles
-
Sustained delivery of sphingosine-1-phosphate using poly(lactic-co-glycolic acid)-based microparticles stimulates Akt/ERK-eNOS mediated angiogenesis and vascular maturation restoring blood flow in ischemic limbs of mice.Eur J Pharmacol. 2010 May 25;634(1-3):121-31. doi: 10.1016/j.ejphar.2010.02.038. Epub 2010 Mar 3. Eur J Pharmacol. 2010. PMID: 20206620
-
Amphiphilic degradable polymers for immobilization and sustained delivery of sphingosine 1-phosphate.Acta Biomater. 2014 Jul;10(7):3079-90. doi: 10.1016/j.actbio.2014.02.051. Epub 2014 Mar 12. Acta Biomater. 2014. PMID: 24631657 Free PMC article.
-
Local delivery of FTY720 accelerates cranial allograft incorporation and bone formation.Cell Tissue Res. 2012 Mar;347(3):553-66. doi: 10.1007/s00441-011-1217-3. Epub 2011 Aug 24. Cell Tissue Res. 2012. PMID: 21863314 Free PMC article.
-
Lysophosphatidic Acid and Sphingosine-1-Phosphate: A Concise Review of Biological Function and Applications for Tissue Engineering.Tissue Eng Part B Rev. 2015 Dec;21(6):531-42. doi: 10.1089/ten.TEB.2015.0107. Epub 2015 Jul 14. Tissue Eng Part B Rev. 2015. PMID: 26035484 Free PMC article. Review.
-
Sphingosine 1-phosphate (S1P) signalling: Role in bone biology and potential therapeutic target for bone repair.Pharmacol Res. 2017 Nov;125(Pt B):232-245. doi: 10.1016/j.phrs.2017.08.013. Epub 2017 Sep 22. Pharmacol Res. 2017. PMID: 28855094 Free PMC article. Review.
Cited by
-
Enhanced osseous integration of human trabecular allografts following surface modification with bioactive lipids.Drug Deliv Transl Res. 2016 Apr;6(2):96-104. doi: 10.1007/s13346-015-0244-0. Drug Deliv Transl Res. 2016. PMID: 26169381 Free PMC article.
-
Alginate hydrogels of varied molecular weight distribution enable sustained release of sphingosine-1-phosphate and promote angiogenesis.J Biomed Mater Res A. 2018 Jan;106(1):138-146. doi: 10.1002/jbm.a.36217. Epub 2017 Sep 26. J Biomed Mater Res A. 2018. PMID: 28875559 Free PMC article.
-
Delivery of S1P receptor-targeted drugs via biodegradable polymer scaffolds enhances bone regeneration in a critical size cranial defect.J Biomed Mater Res A. 2014 Apr;102(4):1210-8. doi: 10.1002/jbm.a.34779. Epub 2013 Oct 17. J Biomed Mater Res A. 2014. PMID: 23640833 Free PMC article.
-
Dual Affinity Heparin-Based Hydrogels Achieve Pro-Regenerative Immunomodulation and Microvascular Remodeling.ACS Biomater Sci Eng. 2018 Apr 9;4(4):1241-1250. doi: 10.1021/acsbiomaterials.6b00706. Epub 2017 Feb 20. ACS Biomater Sci Eng. 2018. PMID: 29682605 Free PMC article.
-
Spatially localized recruitment of anti-inflammatory monocytes by SDF-1α-releasing hydrogels enhances microvascular network remodeling.Biomaterials. 2016 Jan;77:280-90. doi: 10.1016/j.biomaterials.2015.10.045. Epub 2015 Oct 23. Biomaterials. 2016. PMID: 26613543 Free PMC article.
References
-
- Griffith CK, Miller C, Sainson RCA. Diffusion limits of an in vitro thick prevascularized tissue. Tissue Eng. 2005;11(1–2):257–66. - PubMed
-
- Wieghaus KA, Capitosti SM, Anderson CR, Price RJ, Blackman BB, Brown ML, et al. Small molecule inducers of angiogenesis for tissue engineering. Tissue Eng. 2006;12(7):1903–13. - PubMed
-
- Botchwey EA, Dupree MA, Pollack SR, Levine EM, Laurencin CT. Tissue engineered bone: measurement of nutrient transport in three-dimensional matrices. J Biomed Mater Res A. 2003;67(1):357–67. - PubMed
-
- Hershey JC, Baskin EP, Corcoran HA, Bett A, Dougherty NM, Gilberto DB, et al. Vascular endothelial growth factor stimulates angiogenesis without improving collateral blood flow following hindlimb ischemia in rabbits. Heart and Vessels. 2003;18(3):142–9. - PubMed
-
- Ziche M, Donnini S, Morbidelli L. Development of new drugs in angiogenesis. Curr Drug Targets. 2004;5(5):485–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

