Inactivation of lsr2 results in a hypermotile phenotype in Mycobacterium smegmatis
- PMID: 18408023
- PMCID: PMC2446759
- DOI: 10.1128/JB.00023-08
Inactivation of lsr2 results in a hypermotile phenotype in Mycobacterium smegmatis
Abstract
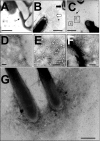
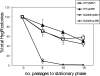
Mycobacterial species are characterized by the presence of lipid-rich, hydrophobic cell envelopes. These cell envelopes contribute to properties such as roughness of colonies, aggregation of cells in liquid culture without detergent, and biofilm formation. We describe here a mutant strain of Mycobacterium smegmatis, called DL1215, which demonstrates marked deviations from the above-mentioned phenotypes. DL1215 arose spontaneously from a strain deficient for the stringent response (M. smegmatis Delta rel(Msm) strain) and is not a reversion to a wild-type phenotype. The nature of the spontaneous mutation was a single base-pair deletion in the lsr2 gene, leading to the formation of a truncated protein product. The DL1215 strain was complicated by having both inactivated rel(Msm) and lsr2 genes, and so a single lsr2 mutant was created to analyze the gene's function. The lsr2 gene was inactivated in the wild-type M. smegmatis mc(2)155 strain by allelic replacement to create strain DL2008. Strain DL2008 shows characteristics unique from those of both the wild-type and Delta rel(Msm) strains, some of which include a greatly enhanced ability to slide over agar surfaces (referred to here as "hypermotility"), greater resistance to phage infection and to the antibiotic kanamycin, and an inability to form biofilms. Complementation of the DL2008 mutant with a plasmid containing lsr2 (pLSR2) reverts the strain to the mc(2)155 phenotype. Although these phenotypic differences allude to changes in cell surface lipids, no difference is observed in glycopeptidolipids, polar lipids, apolar lipids, or mycolic acids of the cell wall.
Figures






Similar articles
-
Roles of Lsr2 in colony morphology and biofilm formation of Mycobacterium smegmatis.J Bacteriol. 2006 Jan;188(2):633-41. doi: 10.1128/JB.188.2.633-641.2006. J Bacteriol. 2006. PMID: 16385053 Free PMC article.
-
Lsr2 and Its Novel Paralogue Mediate the Adjustment of Mycobacterium smegmatis to Unfavorable Environmental Conditions.mSphere. 2021 May 12;6(3):e00290-21. doi: 10.1128/mSphere.00290-21. mSphere. 2021. PMID: 33980681 Free PMC article.
-
Transcriptional regulation of multi-drug tolerance and antibiotic-induced responses by the histone-like protein Lsr2 in M. tuberculosis.PLoS Pathog. 2007 Jun;3(6):e87. doi: 10.1371/journal.ppat.0030087. PLoS Pathog. 2007. PMID: 17590082 Free PMC article.
-
Defining a temporal order of genetic requirements for development of mycobacterial biofilms.Mol Microbiol. 2017 Sep;105(5):794-809. doi: 10.1111/mmi.13734. Epub 2017 Jul 7. Mol Microbiol. 2017. PMID: 28628249 Free PMC article.
-
The relA homolog of Mycobacterium smegmatis affects cell appearance, viability, and gene expression.J Bacteriol. 2005 Apr;187(7):2439-47. doi: 10.1128/JB.187.7.2439-2447.2005. J Bacteriol. 2005. PMID: 15774887 Free PMC article.
Cited by
-
A nucleoid-associated protein is involved in the emergence of antibiotic resistance by promoting the frequent exchange of the replicative DNA polymerase in M. smegmatis.bioRxiv [Preprint]. 2024 Jan 3:2023.06.12.544663. doi: 10.1101/2023.06.12.544663. bioRxiv. 2024. Update in: mSphere. 2024 May 29;9(5):e0012224. doi: 10.1128/msphere.00122-24. PMID: 38260554 Free PMC article. Updated. Preprint.
-
Tale of Twin Bifunctional Second Messenger (p)ppGpp Synthetases and Their Function in Mycobacteria.ACS Omega. 2023 Aug 25;8(36):32258-32270. doi: 10.1021/acsomega.3c03557. eCollection 2023 Sep 12. ACS Omega. 2023. PMID: 37720788 Free PMC article. Review.
-
Rv1717 Is a Cell Wall - Associated β-Galactosidase of Mycobacterium tuberculosis That Is Involved in Biofilm Dispersion.Front Microbiol. 2021 Jan 15;11:611122. doi: 10.3389/fmicb.2020.611122. eCollection 2020. Front Microbiol. 2021. PMID: 33584576 Free PMC article.
-
H-NS plays a role in expression of Acinetobacter baumannii virulence features.Infect Immun. 2013 Jul;81(7):2574-83. doi: 10.1128/IAI.00065-13. Epub 2013 May 6. Infect Immun. 2013. PMID: 23649094 Free PMC article.
-
Stringent Response in Mycobacteria: From Biology to Therapeutic Potential.Pathogens. 2021 Nov 1;10(11):1417. doi: 10.3390/pathogens10111417. Pathogens. 2021. PMID: 34832573 Free PMC article. Review.
References
-
- Alexander, D. C., J. R. Jones, T. Tan, J. M. Chen, and J. Lui. 2004. PimF, a mannosyltransferase of mycobacteria, is involved in the biosynthesis of phosphatidylinositol mannosides and lipoarabinomannan. J. Biol. Chem. 27918824-18833. - PubMed
-
- Bardarov, S., S. Bardarov, Jr., M. S. Pavelka, Jr., V. Sambandamurthy, M. Larsen, J. Tufariello, J. Chan, G. Hatfull, and W. R. Jacobs, Jr. 2002. Specialized transduction: an efficient method for generating marked and unmarked targeted gene disruptions in Mycobacterium tuberculosis, M. bovis BCG and M. smegmatis. Microbiology 1483007-3017. - PubMed
-
- Reference deleted.
-
- Butcher, P. D., J. A. Mangan, and I. M. Monahan. 1998. Intracellular gene expression, p. 285-306. In T. Parish and N. G. Stoker (ed.), Mycobacterial Protocols, 1st ed. Humana Press, Totowa, NJ.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

