Microwave-induced nucleophilic [18F]fluorination on aromatic rings: synthesis and effect of halogen on [18F]fluoride substitution of meta-halo (F, Cl, Br, I)-benzonitrile derivatives
- PMID: 18417350
- PMCID: PMC2553010
- DOI: 10.1016/j.apradiso.2008.03.003
Microwave-induced nucleophilic [18F]fluorination on aromatic rings: synthesis and effect of halogen on [18F]fluoride substitution of meta-halo (F, Cl, Br, I)-benzonitrile derivatives
Abstract
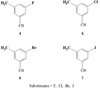
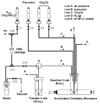
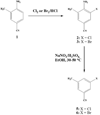
The meta-halo-3-methylbenzonitrile derivatives (-F, -Cl, -Br, -I) were synthesized as model compounds to study reactivity towards aromatic nucleophilic substitution. A single-mode microwave system was incorporated into a commercial radiochemical synthetic module for (18)F labeling. Labeling yields of 64% for fluoro-, 13% for bromo- and 9% for chloro-precursors were achieved in DMSO in <3 min. The observed order of reactivity of the leaving groups toward aromatic nucleophilic substitution was F>>Br>Cl>>>I.
Figures




Similar articles
-
Design and synthesis of a new [18F]fluoropyridine-based haloacetamide reagent for the labeling of oligonucleotides: 2-bromo-N-[3-(2-[18F]fluoropyridin-3-yloxy)propyl]acetamide.Bioconjug Chem. 2004 May-Jun;15(3):617-27. doi: 10.1021/bc049979u. Bioconjug Chem. 2004. PMID: 15149190
-
Electrochemical radiofluorination. Part 2. Anodic monofluorination of substituted benzenes using [18F]fluoride.Appl Radiat Isot. 2003 Jun;58(6):679-83. doi: 10.1016/s0969-8043(03)00093-9. Appl Radiat Isot. 2003. PMID: 12798377
-
Automated synthesis and purification of [18F]fluoro-[di-deutero]methyl tosylate.J Labelled Comp Radiopharm. 2013 Jun 15;56(7):360-3. doi: 10.1002/jlcr.3043. Epub 2013 Apr 17. J Labelled Comp Radiopharm. 2013. PMID: 24285440
-
Fluorine-18-labelled fluoropyridines: advances in radiopharmaceutical design.Curr Pharm Des. 2005;11(25):3221-35. doi: 10.2174/138161205774424645. Curr Pharm Des. 2005. PMID: 16250851 Review.
-
Isotope Exchange-Based 18F-Labeling Methods.Bioconjug Chem. 2023 Jan 18;34(1):140-161. doi: 10.1021/acs.bioconjchem.2c00530. Epub 2023 Jan 3. Bioconjug Chem. 2023. PMID: 36594786 Review.
Cited by
-
Automated Synthesis of [18F]Flumazenil Application in GABAA Receptor Neuroimaging Availability for Rat Model of Anxiety.Pharmaceuticals (Basel). 2023 Mar 9;16(3):417. doi: 10.3390/ph16030417. Pharmaceuticals (Basel). 2023. PMID: 36986516 Free PMC article.
-
Molecular imaging probe development: a chemistry perspective.Am J Nucl Med Mol Imaging. 2012 Jul 10;2(3):273-306. Am J Nucl Med Mol Imaging. 2012. PMID: 22943038 Free PMC article.
-
Syntheses of meta-[F]Fluorobenzaldehyde and meta-[F]Fluorobenzylbromide from Phenyl(3-Formylphenyl) Iodonium Salt Precursors.J Labelled Comp Radiopharm. 2011 Apr;54(4):224-228. doi: 10.1002/jlcr.1853. J Labelled Comp Radiopharm. 2011. PMID: 21532942 Free PMC article.
-
Microwave-assisted one-pot synthesis of N-succinimidyl-4[ ¹⁸F]fluorobenzoate ([¹⁸F]SFB).J Vis Exp. 2011 Jun 28;(52):2755. doi: 10.3791/2755. J Vis Exp. 2011. PMID: 21730951 Free PMC article.
-
Microfluidics: a groundbreaking technology for PET tracer production?Molecules. 2013 Jul 5;18(7):7930-56. doi: 10.3390/molecules18077930. Molecules. 2013. PMID: 23884128 Free PMC article. Review.
References
-
- Al-Labadi A, Zeller K-P, Machulla H-J. Synthesis of 6-[18F]fluoroveratraldehyde by nucleophilic halogen exchange at electron-rich precursors. J. Radioanal. Nuc. Chem. 2006;270(2):313–318.
-
- Arvela RK, Leadbeater NE. Fast and easy halide exchange in aryl halides. Synlett. 2003;8:1145–1148.
-
- Couturier O, Luxen A, Chatal J-F, Vuillez J-P, Rigo P, Hustinx R. Fluorinated tracers for imaging cancer with positron emission tomography. Eur. J. Nucl. Med. & Mol. Imaging. 2004;31(8):1182–1206. - PubMed
-
- de Vries EFJ, Waarde AV, Buursma AR, Vaalburg W. Synthesis and in vivo evaluation of 18F-desbromo-DuP-697 as a PET tracer for cyclooxygenase-2 expression. J. Nuc. Med. 2003;44(10):1700–1706. - PubMed
-
- Ding Y-S. 18F-Labeled biomolecules for PET studies in the neurosciences. J. Fluorine Chem. 2000;101:291–295.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

