Inhibitory interactions among olfactory glomeruli do not necessarily reflect spatial proximity
- PMID: 18417626
- PMCID: PMC2525721
- DOI: 10.1152/jn.90231.2008
Inhibitory interactions among olfactory glomeruli do not necessarily reflect spatial proximity
Abstract
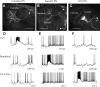
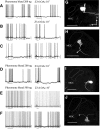
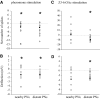
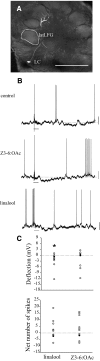
Inhibitory interactions shape the activity of output neurons in primary olfactory centers and promote contrast enhancement of odor representations. Patterns of interglomerular connectivity, however, are largely unknown. To test whether the proximity of glomeruli to one another is correlated with interglomerular inhibitory interactions, we used intracellular recording and staining methods to record the responses of projection (output) neurons (PNs) associated with glomeruli of known olfactory tuning in the primary olfactory center of the moth Manduca sexta. We focused on Toroid I, a glomerulus in the male-specific macroglomerular complex (MGC) specialized to one of the two key components of the conspecific females' sex pheromone, and the adjacent, sexually isomorphic glomerulus 35, which is highly sensitive to Z-3-hexenyl acetate (Z3-6:OAc). We used the two odorants to activate these reference glomeruli and tested the effects of olfactory activation in other glomeruli. We found that Toroid-I PNs were not inhibited by input to G35, whereas G35 PNs were inhibited by input to Toroid-I PNs. We also recorded the responses of PNs arborizing in other sexually isomorphic glomeruli to stimulation with the sex pheromone and Z3-6:OAc. We found that inhibitory responses were not related to proximity to the MGC and G35: both distant and adjacent PNs were inhibited by stimulation with the sex pheromone, some others were affected by only one odorant, and yet others by neither. Similar results were obtained in female PNs recorded in proximity to female-specific glomeruli. Our findings indicate that inhibitory interactions among glomeruli are widespread and independent of their spatial proximity.
Figures

Similar articles
-
Glomerular interactions in olfactory processing channels of the antennal lobes.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2013 Nov;199(11):929-46. doi: 10.1007/s00359-013-0842-6. Epub 2013 Jul 28. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2013. PMID: 23893248 Free PMC article. Review.
-
Chemosensory selectivity of output neurons innervating an identified, sexually isomorphic olfactory glomerulus.J Neurosci. 2005 Aug 31;25(35):8017-26. doi: 10.1523/JNEUROSCI.1314-05.2005. J Neurosci. 2005. PMID: 16135759 Free PMC article.
-
Representation of binary pheromone blends by glomerulus-specific olfactory projection neurons.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2004 Dec;190(12):1023-37. doi: 10.1007/s00359-004-0559-7. Epub 2004 Sep 17. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2004. PMID: 15378331
-
Local inhibition modulates odor-evoked synchronization of glomerulus-specific output neurons.Nat Neurosci. 2002 Jun;5(6):557-65. doi: 10.1038/nn0602-859. Nat Neurosci. 2002. PMID: 12006983
-
GABAergic mechanisms that shape the temporal response to odors in moth olfactory projection neurons.Ann N Y Acad Sci. 1998 Nov 30;855:475-81. doi: 10.1111/j.1749-6632.1998.tb10608.x. Ann N Y Acad Sci. 1998. PMID: 9929641 Review.
Cited by
-
Coincidence of pheromone and plant odor leads to sensory plasticity in the heliothine olfactory system.PLoS One. 2017 May 3;12(5):e0175513. doi: 10.1371/journal.pone.0175513. eCollection 2017. PLoS One. 2017. PMID: 28467500 Free PMC article.
-
Glomerular interactions in olfactory processing channels of the antennal lobes.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2013 Nov;199(11):929-46. doi: 10.1007/s00359-013-0842-6. Epub 2013 Jul 28. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2013. PMID: 23893248 Free PMC article. Review.
-
Neuronal processing of complex mixtures establishes a unique odor representation in the moth antennal lobe.Front Neural Circuits. 2011 May 11;5:7. doi: 10.3389/fncir.2011.00007. eCollection 2011. Front Neural Circuits. 2011. PMID: 21772814 Free PMC article.
-
Serotonin modulates olfactory processing in the antennal lobe of Drosophila.J Neurogenet. 2009;23(4):366-77. doi: 10.3109/01677060903085722. J Neurogenet. 2009. PMID: 19863268 Free PMC article.
-
Reconstruction of virtual neural circuits in an insect brain.Front Neurosci. 2009 Sep 15;3(2):206-13. doi: 10.3389/neuro.01.028.2009. eCollection 2009 Sep. Front Neurosci. 2009. PMID: 20011143 Free PMC article.
References
-
- Abel R, Rybak J, Menzel R. Structure and response patterns of olfactory interneurons in the honeybee, Apis mellifera. J Comp Neurol 437: 363–383, 2001. - PubMed
-
- Aungst JL, Heyward PM, Puche AC, Karnup SV, Hayar A, Szabo G, Shipley MT. Centre-surround inhibition among olfactory bulb glomeruli. Nature 426: 623–629, 2003. - PubMed
-
- Berkowicz DA, Trombley PQ, Shepherd GM. Evidence for glutamate as the olfactory receptor cell neurotransmitter. J Neurophysiol 71: 2557–2561, 1994. - PubMed
-
- Boeckh J, Tolbert LP. Synaptic organization and development of the antennal lobe in insects. Microscop Res Techn 24: 260–280, 1993. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

