Structural basis of toll-like receptor 3 signaling with double-stranded RNA
- PMID: 18420935
- PMCID: PMC2761030
- DOI: 10.1126/science.1155406
Structural basis of toll-like receptor 3 signaling with double-stranded RNA
Abstract
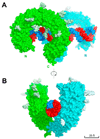
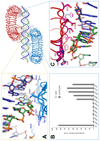
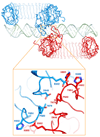
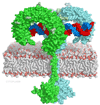
Toll-like receptor 3 (TLR3) recognizes double-stranded RNA (dsRNA), a molecular signature of most viruses, and triggers inflammatory responses that prevent viral spread. TLR3 ectodomains (ECDs) dimerize on oligonucleotides of at least 40 to 50 base pairs in length, the minimal length required for signal transduction. To establish the molecular basis for ligand binding and signaling, we determined the crystal structure of a complex between two mouse TLR3-ECDs and dsRNA at 3.4 angstrom resolution. Each TLR3-ECD binds dsRNA at two sites located at opposite ends of the TLR3 horseshoe, and an intermolecular contact between the two TLR3-ECD C-terminal domains coordinates and stabilizes the dimer. This juxtaposition could mediate downstream signaling by dimerizing the cytoplasmic Toll interleukin-1 receptor (TIR) domains. The overall shape of the TLR3-ECD does not change upon binding to dsRNA.
Figures
References
-
- Kaisho T, Akira S. J. Allergy Clin. Immunol. 2006;117:979. - PubMed
-
- Iwasaki A, Medzhitov R. Nat. Immunol. 2004;5:987. - PubMed
-
- Gay NJ, Gangloff M. Annu. Rev. Biochem. 2007;76:141. - PubMed
-
- Alexopoulou L, Holt AC, Medzhitov R, Flavell RA. Nature. 2001;413:732. - PubMed
-
- Oshiumi H, Matsumoto M, Funami K, Akazawa T, Seya T. Nat. Immunol. 2003;4:161. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

