Risedronate prevents bone loss in breast cancer survivors: a 2-year, randomized, double-blind, placebo-controlled clinical trial
- PMID: 18427147
- PMCID: PMC3992822
- DOI: 10.1200/JCO.2007.15.2967
Risedronate prevents bone loss in breast cancer survivors: a 2-year, randomized, double-blind, placebo-controlled clinical trial
Abstract
Purpose: Limited data are available on the efficacy of oral bisphosphonate therapy in breast cancer survivors. Our goal was to examine prevention of breast cancer-related bone loss in this cohort.
Patients and methods: Eighty-seven postmenopausal women after chemotherapy for breast cancer were randomly assigned to once-weekly risedronate 35 mg or placebo for 24 months. Outcomes included bone mineral density (BMD) and turnover markers.
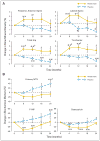
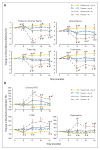
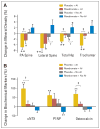
Results: At study initiation, 13% of patients were on an aromatase inhibitor (AI). After 24 months, there were differences of 1.6 to 2.5% (P < .05) at the spine and hip BMD between the placebo and risedronate groups. At study completion, 44% were on an AI. Adjusting for an AI, women on placebo plus AI had a decrease in BMD of (mean +/- SE) 4.8% +/- 0.8% at the spine and 2.8% +/- 0.5% at the total hip (both P < .001). In women on risedronate + AI, the spine decreased by 2.4% +/- 1.1% (P < .05) and was stable at the hip. Women in the placebo group not on an AI, maintained BMD at the spine, and had a 1.2% +/- 0.5% loss at the total hip (P < .05). Women who received risedronate but no AI had the greatest improvement in BMD of 2.2% +/- 0.9% (P < .05) at the total hip. Bone turnover was reduced with risedronate. There were no differences in adverse events between the groups.
Conclusion: We conclude that in postmenopausal women with breast cancer with or without AI therapy, once-weekly oral risedronate was beneficial for spine and hip BMD, reduced bone turnover, and was well tolerated.
Trial registration: ClinicalTrials.gov NCT00118508.
Conflict of interest statement
Although all authors completed the disclosure declaration, the following author(s) indicated a financial or other interest that is relevant to the subject matter under consideration in this article. Certain relationships marked with a “U” are those for which no compensation was received; those relationships marked with a “C” were compensated. For a detailed description of the disclosure categories, or for more information about ASCO’s conflict of interest policy, please refer to the Author Disclosure Declaration and the Disclosures of Potential Conflicts of Interest section in Information for Contributors.
Figures
References
-
- Early Breast Cancer Trialists’ Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet. 2005;365:1687–1717. - PubMed
-
- Pfeilschifter J, Diel IJ. Osteoporosis due to cancer treatment: Pathogenesis and management. J Clin Oncol. 2000;18:1570–1593. - PubMed
-
- del Mastro L, Venturini M, Sertoli MR, et al. Amenorrhea induced by adjuvant chemotherapy in early breast cancer patients: Prognostic role and clinical implications. Br Cancer Res Treat. 1997;43:183–190. - PubMed
-
- Schilsky RL, Lewis BJ, Sherins RJ, et al. Gonadal dysfunction in patients receiving chemotherapy for cancer. Ann Intern Med. 1980;93:109–116. - PubMed
-
- Shapiro CL, Manola J, LeBoff M. Ovarian failure after adjuvant chemotherapy is associated with rapid bone loss in women with early-stage breast cancer. J Clin Oncol. 2001;19:3306–3311. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

