Resveratrol induces apoptosis in K562 (chronic myelogenous leukemia) cells by targeting a key survival protein, heat shock protein 70
- PMID: 18429957
- PMCID: PMC11159327
- DOI: 10.1111/j.1349-7006.2008.00809.x
Resveratrol induces apoptosis in K562 (chronic myelogenous leukemia) cells by targeting a key survival protein, heat shock protein 70
Abstract
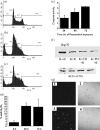
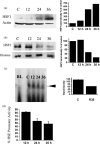
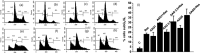
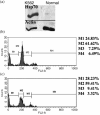
Chronic myelogenous leukemia (CML) is a myeloproliferative disease associated with a characteristic chromosomal translocation called the Philadelphia chromosome. This results in the expression of the Bcr-Abl fusion protein, a constitutively active protein tyrosine kinase. Although there are a few treatment options with Bcr-Abl kinase inhibitors, drug resistance is often encountered. One of the major obstacles in overcoming drug resistance in CML is the high endogenous levels of heat shock protein 70 (Hsp70). Resveratrol is a phytoalexin produced by several plants. We studied the chemotherapeutic effects and mode of action of resveratrol on K562 (CML) cells. Resveratrol induced apoptosis in K562 cells in a time-dependent manner. This was established by increased annexin V binding, corroborated with an enhanced caspase-3 activity and a rise in the sub-G(0)/G(1) population. Resveratrol treatment also caused suppression of Hsp70 both in mRNA and protein levels. The downregulation of Hsp70 by resveratrol exposure was correlated with a diminished presence of heat shock factor 1 (HSF1) in the nucleus, and the downregulation of transcriptional activity of HSF1. High endogenous levels of Hsp70 have been found to be a deterrent for sensitivity to chemotherapy. We show here that resveratrol could considerably enhance the apoptosis induction in K562 cells by 17-allylamino-17-demethoxygeldanamycin, an anticancer agent that inhibits Hsp90 but augments Hsp70 levels. We conclude that resveratrol significantly downregulated Hsp70 levels through inhibition of HSF1 transcriptional activity and appreciably augmented the pro-apoptotic effects of 17-allylamino-17-demethoxygeldanamycin.
Figures



Similar articles
-
Modulation of Akt and ERK1/2 pathways by resveratrol in chronic myelogenous leukemia (CML) cells results in the downregulation of Hsp70.PLoS One. 2010 Jan 14;5(1):e8719. doi: 10.1371/journal.pone.0008719. PLoS One. 2010. PMID: 20090934 Free PMC article.
-
Genomic mechanisms of p210BCR-ABL signaling: induction of heat shock protein 70 through the GATA response element confers resistance to paclitaxel-induced apoptosis.J Biol Chem. 2004 Aug 20;279(34):35604-15. doi: 10.1074/jbc.M401851200. Epub 2004 May 21. J Biol Chem. 2004. PMID: 15155749
-
Abrogation of heat shock protein 70 induction as a strategy to increase antileukemia activity of heat shock protein 90 inhibitor 17-allylamino-demethoxy geldanamycin.Cancer Res. 2005 Nov 15;65(22):10536-44. doi: 10.1158/0008-5472.CAN-05-1799. Cancer Res. 2005. PMID: 16288046
-
Heat shock proteins in multiple myeloma.Oncotarget. 2014 Mar 15;5(5):1132-48. doi: 10.18632/oncotarget.1584. Oncotarget. 2014. PMID: 24675290 Free PMC article. Review.
-
The anticancer effects of resveratrol: modulation of transcription factors.Nutr Cancer. 2012;64(4):493-502. doi: 10.1080/01635581.2012.667862. Epub 2012 Apr 6. Nutr Cancer. 2012. PMID: 22482424 Free PMC article. Review.
Cited by
-
Resveratrol inhibits proliferation and promotes apoptosis of neuroblastoma cells: role of sirtuin 1.Neurochem Res. 2011 Feb;36(2):187-94. doi: 10.1007/s11064-010-0296-y. Epub 2010 Oct 24. Neurochem Res. 2011. PMID: 20972827
-
Resveratrol protects leukemic cells against cytotoxicity induced by proteasome inhibitors via induction of FOXO1 and p27Kip1.BMC Cancer. 2011 Mar 19;11:99. doi: 10.1186/1471-2407-11-99. BMC Cancer. 2011. PMID: 21418583 Free PMC article.
-
Inducible hsp70 in the regulation of cancer cell survival: analysis of chaperone induction, expression and activity.Cancers (Basel). 2011 Oct 21;3(4):3921-56. doi: 10.3390/cancers3043921. Cancers (Basel). 2011. PMID: 24213118 Free PMC article.
-
Cellular and Molecular Targets of Resveratrol on Lymphoma and Leukemia Cells.Molecules. 2017 May 27;22(6):885. doi: 10.3390/molecules22060885. Molecules. 2017. PMID: 28555002 Free PMC article. Review.
-
Benzo(a)pyrene Induced p53 Mediated Male Germ Cell Apoptosis: Synergistic Protective Effects of Curcumin and Resveratrol.Front Pharmacol. 2016 Aug 8;7:245. doi: 10.3389/fphar.2016.00245. eCollection 2016. Front Pharmacol. 2016. PMID: 27551266 Free PMC article.
References
-
- Deininger MWN, Goldman JM, Melo JV. The molecular biology of chronic myeloid leukemia. Blood 2000; 96: 3343–56. - PubMed
-
- Bedi A, Barber JP, Bedi GC et al . BCR‐ABL‐mediated inhibition of apoptosis with delay of G2/M transition after DNA damage: a mechanism of resistance to multiple anticancer agents. Blood 1995; 86: 1148–58. - PubMed
-
- Amarante‐Mendes GP, Kim CN, Liu L et al . Bcr‐Abl exerts its antiapoptotic effect against diverse apoptotic stimuli through blockage of mitochondrial release of cytochrome C, activation of caspase‐3. Blood 1998; 91: 1700–5. - PubMed
-
- Kharas MG, Fruman DA. ABL oncogenes and phosphoinositide 3‐kinase: mechanism of activation and downstream effectors. Cancer Res 2005; 65: 2047–53. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

