Effect of cysteine mutagenesis on the function and disulfide bond formation of human ABCG2
- PMID: 18430864
- PMCID: PMC2632310
- DOI: 10.1124/jpet.108.138115
Effect of cysteine mutagenesis on the function and disulfide bond formation of human ABCG2
Abstract
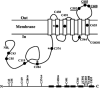
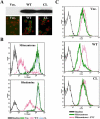
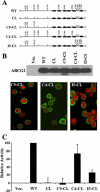
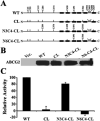
ABCG2 is a member of the ATP-binding cassette (ABC) transporter superfamily. Its overexpression causes multidrug resistance in cancer chemotherapy. Based on its apparent half size in sequence when compared with other traditional ABC transporters, ABCG2 has been thought to exist and function as a homodimer linked by intermolecular disulfide bonds. However, recent evidence suggests that ABCG2 may exist as a higher form of oligomers due to noncovalent interactions. In this study, we attempted to create a cysless mutant ABCG2 as a tool for further characterization of this molecule. However, we found that the cysless mutant ABCG2 is well expressed but not functional. Mapping of the cysteine residues showed that three cysteine residues (Cys284, Cys374, and Cys438) are required concurrently for the function of ABCG2 and potentially for intramolecular disulfide bond formation. We also found that the cysteine residues (Cys592, Cys603, and Cys608) in the third extracellular loop are involved in forming intermolecular disulfide bonds and that mutation of these residues does not affect the expression or drug transport activity of human ABCG2. Thus, we conclude that Cys284, Cys374, and Cys438, which may be involved in intramolecular disulfide bond formation, are concurrently required for ABCG2 function, whereas Cys592, Cys603, and Cys608, potentially involved in intermolecular disulfide bond formation, are not required.
Figures

Similar articles
-
Identification of cysteine residues critically involved in homodimer formation and protein expression of human ATP-binding cassette transporter ABCG2: a new approach using the flp recombinase system.J Exp Ther Oncol. 2006;5(3):205-22. J Exp Ther Oncol. 2006. PMID: 16528971
-
Identification of intra- and intermolecular disulfide bridges in the multidrug resistance transporter ABCG2.J Biol Chem. 2005 Nov 4;280(44):36926-34. doi: 10.1074/jbc.M502937200. Epub 2005 Aug 17. J Biol Chem. 2005. PMID: 16107343
-
Intramolecular disulfide bond is a critical check point determining degradative fates of ATP-binding cassette (ABC) transporter ABCG2 protein.J Biol Chem. 2007 Sep 21;282(38):27841-6. doi: 10.1074/jbc.C700133200. Epub 2007 Aug 8. J Biol Chem. 2007. PMID: 17686774
-
Human ABC transporter ABCG2 in xenobiotic protection and redox biology.Drug Metab Rev. 2006;38(3):371-91. doi: 10.1080/03602530600727947. Drug Metab Rev. 2006. PMID: 16877258 Review.
-
Quality control of human ABCG2 protein in the endoplasmic reticulum: ubiquitination and proteasomal degradation.Adv Drug Deliv Rev. 2009 Jan 31;61(1):66-72. doi: 10.1016/j.addr.2008.08.008. Epub 2008 Dec 11. Adv Drug Deliv Rev. 2009. PMID: 19111842 Review.
Cited by
-
Dimerization of ABCG2 analysed by bimolecular fluorescence complementation.PLoS One. 2011;6(10):e25818. doi: 10.1371/journal.pone.0025818. Epub 2011 Oct 3. PLoS One. 2011. PMID: 21991363 Free PMC article.
-
Different roles of TM5, TM6, and ECL3 in the oligomerization and function of human ABCG2.Biochemistry. 2012 May 1;51(17):3634-41. doi: 10.1021/bi300301a. Epub 2012 Apr 19. Biochemistry. 2012. PMID: 22497316 Free PMC article.
-
Cancer drug resistance: redox resetting renders a way.Oncotarget. 2016 Jul 5;7(27):42740-42761. doi: 10.18632/oncotarget.8600. Oncotarget. 2016. PMID: 27057637 Free PMC article. Review.
-
3D structure of the transporter ABCG2-What's new?Br J Pharmacol. 2020 Apr;177(7):1485-1496. doi: 10.1111/bph.14991. Epub 2020 Feb 11. Br J Pharmacol. 2020. PMID: 31985041 Free PMC article. Review.
-
Redox-sensitive DNA binding by homodimeric Methanosarcina acetivorans MsvR is modulated by cysteine residues.BMC Microbiol. 2013 Jul 16;13:163. doi: 10.1186/1471-2180-13-163. BMC Microbiol. 2013. PMID: 23865844 Free PMC article.
References
-
- Allen JD, Schinkel AH. Multidrug resistance and pharmacological protection mediated by the breast cancer resistance protein (BCRP/ABCG2). Mol Cancer Ther. 2002;1:427–434. - PubMed
-
- Bakos E, Evers R, Calenda G, Tusnady GE, Szakacs G, Varadi A, Sarkadi B. Characterization of the amino-terminal regions in the human multidrug resistance protein (MRP1). J Cell Sci. 2000;113:4451–4461. - PubMed
-
- Bhatia A, Schafer HJ, Hrycyna CA. Oligomerization of the human ABC transporter ABCG2: evaluation of the native protein and chimeric dimers. Biochemistry. 2005;44:10893–10904. - PubMed
-
- Denning GM, Anderson MP, Amara JF, Marshall J, Smith AE, Welsh MJ. Processing of mutant cystic fibrosis transmembrane conductance regulator is temperature-sensitive. Nature. 1992;358:761–764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

