A yeast TDP-43 proteinopathy model: Exploring the molecular determinants of TDP-43 aggregation and cellular toxicity
- PMID: 18434538
- PMCID: PMC2359814
- DOI: 10.1073/pnas.0802082105
A yeast TDP-43 proteinopathy model: Exploring the molecular determinants of TDP-43 aggregation and cellular toxicity
Abstract
Protein misfolding is intimately associated with devastating human neurodegenerative diseases, including Alzheimer's, Huntington's, and Parkinson's. Although disparate in their pathophysiology, many of these disorders share a common theme, manifested in the accumulation of insoluble protein aggregates in the brain. Recently, the major disease protein found in the pathological inclusions of two of these diseases, amyotrophic lateral sclerosis (ALS) and frontal temporal lobar degeneration with ubiquitin-positive inclusions (FTLD-U), was identified as the 43-kDa TAR-DNA-binding protein (TDP-43), providing a molecular link between them. TDP-43 is a ubiquitously expressed nuclear protein that undergoes a pathological conversion to an aggregated cytoplasmic localization in affected regions of the nervous system. Whether TDP-43 itself can convey toxicity and whether its abnormal aggregation is a cause or consequence of pathogenesis remain unknown. We report a yeast model to define mechanisms governing TDP-43 subcellular localization and aggregation. Remarkably, this simple model recapitulates several salient features of human TDP-43 proteinopathies, including conversion from nuclear localization to cytoplasmic aggregation. We establish a connection between this aggregation and toxicity. The pathological features of TDP-43 are distinct from those of yeast models of other protein-misfolding diseases, such as polyglutamine. This suggests that the yeast model reveals specific aspects of the underlying biology of the disease protein rather than general cellular stresses associated with accumulating misfolded proteins. This work provides a mechanistic framework for investigating the toxicity of TDP-43 aggregation relevant to human disease and establishes a manipulable, high-throughput model for discovering potential therapeutic strategies.
Conflict of interest statement
Conflict of interest statement: S.L. is a cofounder of and owns stock in FoldRx Pharmaceuticals, a company developing therapies for diseases of protein misfolding. S.L. and A.D.G. are inventors on patents and patent applications that have been licensed to FoldRx Pharmaceuticals.
Figures
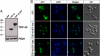

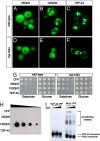

References
-
- Dobson CM. Protein folding and misfolding. Nature. 2003;426:884–890. - PubMed
-
- Forman MS, Trojanowski JQ, Lee VM. Neurodegenerative diseases: A decade of discoveries paves the way for therapeutic breakthroughs. Nat Med. 2004;10:1055–1063. - PubMed
-
- Lee VM, Balin BJ, Otvos L, Jr, Trojanowski JQ. A68: A major subunit of paired helical filaments and derivatized forms of normal Tau. Science. 1991;251:675–678. - PubMed
-
- Glenner GG, Wong CW. Alzheimer's disease: Initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun. 1984;120:885–890. - PubMed
-
- Spillantini MG, et al. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–840. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

