The role of mineralocorticoid receptor expression in brain remodeling after cerebral ischemia
- PMID: 18436714
- PMCID: PMC2488212
- DOI: 10.1210/en.2007-1770
The role of mineralocorticoid receptor expression in brain remodeling after cerebral ischemia
Abstract
Mineralocorticoid receptors (MRs) are classically known to be expressed in the distal collecting duct of the kidney. Recently it was reported that MR is identified in the heart and vasculature. Although MR expression is also found in the brain, it is restricted to the hippocampus and cerebral cortex under normal condition, and the role played by MRs in brain remodeling after cerebral ischemia remains unclear. In the present study, we used the mouse 20-min middle cerebral artery occlusion model to examine the time course of MR expression and activity in the ischemic brain. We found that MR-positive cells remarkably increased in the ischemic striatum, in which MR expression is not observed under normal conditions, during the acute and, especially, subacute phases after stroke and that the majority of MR-expressing cells were astrocytes that migrated to the ischemic core. Treatment with the MR antagonist spironolactone markedly suppressed superoxide production within the infarct area during this period. Quantitative real-time RT-PCR revealed that spironolactone stimulated the expression of neuroprotective or angiogenic factors, such as basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF), whereas immunohistochemical analysis showed astrocytes to be cells expressing bFGF and VEGF. Thereby the incidence of apoptosis was reduced. The up-regulated bFGF and VEGF expression also appeared to promote endogenous angiogenesis and blood flow within the infarct area and to increase the number of neuroblasts migrating toward the ischemic striatum. By these beneficial effects, the infarct volume was significantly reduced in spironolactone-treated mice. Spironolactone may thus provide therapeutic neuroprotective effects in the ischemic brain after stroke.
Figures



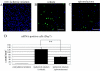





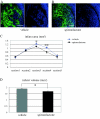
Comment in
-
Stroke therapy: is spironolactone the Holy Grail?Endocrinology. 2008 Aug;149(8):3761-3. doi: 10.1210/en.2008-0724. Endocrinology. 2008. PMID: 18648035 No abstract available.
Similar articles
-
Aldosterone-induced oxidative stress and inflammation in the brain are mediated by the endothelial cell mineralocorticoid receptor.Brain Res. 2016 Apr 15;1637:146-153. doi: 10.1016/j.brainres.2016.02.034. Epub 2016 Feb 26. Brain Res. 2016. PMID: 26923165
-
Myeloid mineralocorticoid receptor during experimental ischemic stroke: effects of model and sex.J Am Heart Assoc. 2012 Oct;1(5):e002584. doi: 10.1161/JAHA.112.002584. Epub 2012 Oct 25. J Am Heart Assoc. 2012. PMID: 23316294 Free PMC article.
-
Intact female stroke-prone hypertensive rats lack responsiveness to mineralocorticoid receptor antagonists.Am J Physiol Regul Integr Comp Physiol. 2007 Oct;293(4):R1754-63. doi: 10.1152/ajpregu.00145.2007. Epub 2007 Aug 1. Am J Physiol Regul Integr Comp Physiol. 2007. PMID: 17670862 Free PMC article.
-
Spironolactone modulates expressions of cardiac mineralocorticoid receptor and 11beta-hydroxysteroid dehydrogenase 2 and prevents ventricular remodeling in post-infarct rat hearts.Hypertens Res. 2007 May;30(5):427-37. doi: 10.1291/hypres.30.427. Hypertens Res. 2007. PMID: 17587755
-
The Role of the Mineralocorticoid Receptor and Mineralocorticoid Receptor-Directed Therapies in Heart Failure.Endocrinology. 2021 Nov 1;162(11):bqab105. doi: 10.1210/endocr/bqab105. Endocrinology. 2021. PMID: 34050730 Review.
Cited by
-
Aldosterone and the mineralocorticoid receptor in the cerebral circulation and stroke.Exp Transl Stroke Med. 2012 Oct 30;4(1):21. doi: 10.1186/2040-7378-4-21. Exp Transl Stroke Med. 2012. PMID: 23110876 Free PMC article.
-
The renin-angiotensin-aldosterone system: a crossroad from arterial hypertension to heart failure.Heart Fail Rev. 2020 Jan;25(1):31-42. doi: 10.1007/s10741-019-09855-5. Heart Fail Rev. 2020. PMID: 31512149 Review.
-
miR-324-5p and miR-30c-2-3p Alter Renal Mineralocorticoid Receptor Signaling under Hypertonicity.Cells. 2022 Apr 19;11(9):1377. doi: 10.3390/cells11091377. Cells. 2022. PMID: 35563683 Free PMC article.
-
Mineralocorticoid Receptor Blockers: Novel Selective Nonsteroidal Mineralocorticoid Receptor Antagonists.Curr Hypertens Rep. 2020 Feb 29;22(3):21. doi: 10.1007/s11906-020-1023-y. Curr Hypertens Rep. 2020. PMID: 32114686 Review.
-
Corticosteroids and perinatal hypoxic-ischemic brain injury.Drug Discov Today. 2018 Oct;23(10):1718-1732. doi: 10.1016/j.drudis.2018.05.019. Epub 2018 May 17. Drug Discov Today. 2018. PMID: 29778695 Free PMC article. Review.
References
-
- Sheppard KE, Autelitano DJ 2002 11β-Hydroxysteroid dehydrogenase 1 transforms 11-dehydrocorticosterone into transcriptionally active glucocorticoid in neonatal rat heart. Endocrinology 143:198–204 - PubMed
-
- Takeda Y, Yoneda T, Demura M, Usukura M, Mabuchi H 2002 Calcineurin inhibition attenuates mineralocorticoid-induced cardiac hypertrophy. Circulation 105:677–679 - PubMed
-
- Brilla CG, Matsubara LS, Weber KT 1993 Anti-aldosterone treatment and the prevention of myocardial fibrosis in primary and secondary hyperaldosteronism. J Mol Cell Cardiol 25:563–575 - PubMed
-
- Kuster GM, Kotlyar E, Rude MK, Siwik DA, Liao R, Colucci WS, Sam F 2005 Mineralocorticoid receptor inhibition ameliorates the transition to myocardial failure and decreases oxidative stress and inflammation in mice with chronic pressure overload. Circulation 111:420–427 - PubMed
-
- Virdis A, Neves MF, Amiri F, Viel E, Touyz RM, Schiffrin EL 2002 Spironolactone improves angiotensin-induced vascular changes and oxidative stress. Hypertension 40:504–510 - PubMed

