Mitochondrial translocation of alpha-synuclein is promoted by intracellular acidification
- PMID: 18440504
- PMCID: PMC2483835
- DOI: 10.1016/j.yexcr.2008.03.012
Mitochondrial translocation of alpha-synuclein is promoted by intracellular acidification
Abstract
Mitochondrial dysfunction plays a central role in the selective vulnerability of dopaminergic neurons in Parkinson's disease (PD) and is influenced by both environmental and genetic factors. Expression of the PD protein alpha-synuclein or its familial mutants often sensitizes neurons to oxidative stress and to damage by mitochondrial toxins. This effect is thought to be indirect, since little evidence physically linking alpha-synuclein to mitochondria has been reported. Here, we show that the distribution of alpha-synuclein within neuronal and non-neuronal cells is dependent on intracellular pH. Cytosolic acidification induces translocation of alpha-synuclein from the cytosol onto the surface of mitochondria. Translocation occurs rapidly under artificially-induced low pH conditions and as a result of pH changes during oxidative or metabolic stress. Binding is likely facilitated by low pH-induced exposure of the mitochondria-specific lipid cardiolipin. These results imply a direct role for alpha-synuclein in mitochondrial physiology, especially under pathological conditions, and in principle, link alpha-synuclein to other PD genes in regulating mitochondrial homeostasis.
Figures
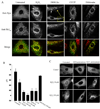
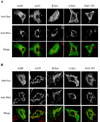

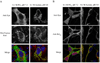
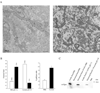

Similar articles
-
Mitochondrial import and accumulation of alpha-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain.J Biol Chem. 2008 Apr 4;283(14):9089-100. doi: 10.1074/jbc.M710012200. Epub 2008 Feb 1. J Biol Chem. 2008. PMID: 18245082 Free PMC article.
-
An in vitro model of Parkinson's disease: linking mitochondrial impairment to altered alpha-synuclein metabolism and oxidative damage.J Neurosci. 2002 Aug 15;22(16):7006-15. doi: 10.1523/JNEUROSCI.22-16-07006.2002. J Neurosci. 2002. PMID: 12177198 Free PMC article.
-
The deglycase activity of DJ-1 mitigates α-synuclein glycation and aggregation in dopaminergic cells: Role of oxidative stress mediated downregulation of DJ-1 in Parkinson's disease.Free Radic Biol Med. 2019 May 1;135:28-37. doi: 10.1016/j.freeradbiomed.2019.02.014. Epub 2019 Feb 20. Free Radic Biol Med. 2019. PMID: 30796974
-
Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse.Free Radic Biol Med. 2013 Sep;62:186-201. doi: 10.1016/j.freeradbiomed.2013.05.042. Epub 2013 Jun 3. Free Radic Biol Med. 2013. PMID: 23743292 Review.
-
A new role for α-synuclein in Parkinson's disease: Alteration of ER-mitochondrial communication.Mov Disord. 2015 Jul;30(8):1026-33. doi: 10.1002/mds.26239. Epub 2015 May 7. Mov Disord. 2015. PMID: 25952565 Review.
Cited by
-
Mitochondrial dysfunction and oxidative stress in Parkinson's disease.Prog Neurobiol. 2013 Jul-Aug;106-107:17-32. doi: 10.1016/j.pneurobio.2013.04.004. Epub 2013 Apr 30. Prog Neurobiol. 2013. PMID: 23643800 Free PMC article. Review.
-
Interactions of amyloidogenic proteins with mitochondrial protein import machinery in aging-related neurodegenerative diseases.Front Physiol. 2023 Nov 2;14:1263420. doi: 10.3389/fphys.2023.1263420. eCollection 2023. Front Physiol. 2023. PMID: 38028797 Free PMC article. Review.
-
α-Synuclein controls mitochondrial calcium homeostasis by enhancing endoplasmic reticulum-mitochondria interactions.J Biol Chem. 2012 May 25;287(22):17914-29. doi: 10.1074/jbc.M111.302794. Epub 2012 Mar 27. J Biol Chem. 2012. PMID: 22453917 Free PMC article.
-
Alpha-Synuclein Toxicity on Protein Quality Control, Mitochondria and Endoplasmic Reticulum.Neurochem Res. 2018 Dec;43(12):2212-2223. doi: 10.1007/s11064-018-2673-x. Epub 2018 Oct 28. Neurochem Res. 2018. PMID: 30370500 Review.
-
Ganglioside lipids accelerate α-synuclein amyloid formation.Biochim Biophys Acta Proteins Proteom. 2018 Aug 2;1866(10):1062-72. doi: 10.1016/j.bbapap.2018.07.004. Online ahead of print. Biochim Biophys Acta Proteins Proteom. 2018. PMID: 30077783 Free PMC article.
References
-
- Abou-Sleiman PM, Muqit MM, Wood NW. Expanding insights of mitochondrial dysfunction in Parkinson's disease. Nat Rev Neurosci. 2006;7:207–219. - PubMed
-
- Lin MT, Beal MF. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 2006;443:787–795. - PubMed
-
- Przedborski S, Tieu K, Perier C, Vila M. MPTP as a mitochondrial neurotoxic model of Parkinson's disease. J Bioenerg Biomembr. 2004;36:375–379. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

