Developmental regulation of NO-mediated VEGF-induced effects in the lung
- PMID: 18441284
- PMCID: PMC2551703
- DOI: 10.1165/rcmb.2007-0024OC
Developmental regulation of NO-mediated VEGF-induced effects in the lung
Abstract
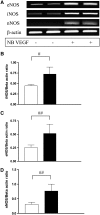
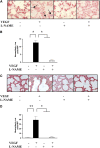
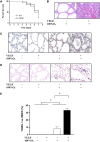
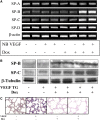
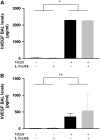
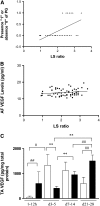
Vascular endothelial growth factor (VEGF) is known to have a pivotal role in lung development and in a variety of pathologic conditions in the adult lung. Our earlier studies have shown that NO is a critical mediator of VEGF-induced vascular and extravascular effects in the adult murine lung. As significant differences have been reported in the cytokine responses in the adult versus the neonatal lung, we hypothesized that there may be significant differences in VEGF-induced alterations in the developing as opposed to the mature lung. Furthermore, nitric oxide (NO) mediation of these VEGF-induced effects may be developmentally regulated. Using a novel externally regulatable lung-targeted transgenic murine model, we found that VEGF-induced pulmonary hemorrhage was mediated by NO-dependent mechanisms in adults and newborns. VEGF enhanced surfactant production in adults as well as increased surfactant and lung development in newborns, via an NO-independent mechanism. While the enhanced survival in hyperoxia in the adult was partly NO-dependent, there was enhanced hyperoxia-induced lung injury in the newborn. In addition, human amniotic fluid VEGF levels correlated positively with surfactant phospholipids. Tracheal aspirate VEGF levels had an initial spike, followed by a decline, and then a subsequent rise, in human neonates with an outcome of bronchopulmonary dysplasia or death. Our data show that VEGF can have injurious as well as potentially beneficial developmental effects, of which some are NO dependent, others NO independent. This opens up the possibility of selective manipulation of any VEGF-based intervention using NO inhibitors for maximal potential clinical benefit.
Figures




Similar articles
-
VEGF causes pulmonary hemorrhage, hemosiderosis, and air space enlargement in neonatal mice.Am J Physiol Lung Cell Mol Physiol. 2004 Jul;287(1):L134-42. doi: 10.1152/ajplung.00050.2004. Epub 2004 Mar 19. Am J Physiol Lung Cell Mol Physiol. 2004. PMID: 15033636
-
Alterations of the thioredoxin system by hyperoxia: implications for alveolar development.Am J Respir Cell Mol Biol. 2009 Nov;41(5):612-9. doi: 10.1165/rcmb.2008-0224OC. Epub 2009 Feb 24. Am J Respir Cell Mol Biol. 2009. PMID: 19244202 Free PMC article.
-
Sexual dimorphism of neonatal lung development.Klin Padiatr. 2013 Mar;225(2):64-69. doi: 10.1055/s-0033-1333758. Epub 2013 Mar 22. Klin Padiatr. 2013. PMID: 23526610 Review.
-
Inhibition of Regulatory-Associated Protein of Mechanistic Target of Rapamycin Prevents Hyperoxia-Induced Lung Injury by Enhancing Autophagy and Reducing Apoptosis in Neonatal Mice.Am J Respir Cell Mol Biol. 2016 Nov;55(5):722-735. doi: 10.1165/rcmb.2015-0349OC. Am J Respir Cell Mol Biol. 2016. PMID: 27374190 Free PMC article.
-
VEGF levels in humans and animal models with RDS and BPD: temporal relationships.Exp Lung Res. 2012 May;38(4):192-203. doi: 10.3109/01902148.2012.663454. Epub 2012 Mar 6. Exp Lung Res. 2012. PMID: 22394267 Free PMC article. Review.
Cited by
-
Current technology in the diagnosis of developmentally related lung disorders.Neonatology. 2012;101(4):353-9. doi: 10.1159/000337356. Epub 2012 Jun 1. Neonatology. 2012. PMID: 22940625 Free PMC article. Review.
-
Role of Nitric Oxide Isoforms in Vascular and Alveolar Development and Lung Injury in Vascular Endothelial Growth Factor Overexpressing Neonatal Mice Lungs.PLoS One. 2016 Jan 22;11(1):e0147588. doi: 10.1371/journal.pone.0147588. eCollection 2016. PLoS One. 2016. PMID: 26799210 Free PMC article.
-
A role for matrix metalloproteinase 9 in IFNγ-mediated injury in developing lungs: relevance to bronchopulmonary dysplasia.Am J Respir Cell Mol Biol. 2011 May;44(5):621-30. doi: 10.1165/rcmb.2010-0058OC. Epub 2011 Jan 7. Am J Respir Cell Mol Biol. 2011. PMID: 21216975 Free PMC article.
-
Increased hyperoxia-induced lung injury in nitric oxide synthase 2 null mice is mediated via angiopoietin 2.Am J Respir Cell Mol Biol. 2012 May;46(5):668-76. doi: 10.1165/rcmb.2011-0074OC. Epub 2012 Jan 6. Am J Respir Cell Mol Biol. 2012. PMID: 22227562 Free PMC article.
-
Renal dopaminergic system: Pathophysiological implications and clinical perspectives.World J Nephrol. 2015 May 6;4(2):196-212. doi: 10.5527/wjn.v4.i2.196. World J Nephrol. 2015. PMID: 25949933 Free PMC article. Review.
References
-
- Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med 2003;9:669–676. - PubMed
-
- Zelzer E, Olsen BR. Multiple roles of vascular endothelial growth factor (VEGF) in skeletal development, growth, and repair. Curr Top Dev Biol 2005;65:169–187. - PubMed
-
- Reynolds LP, Redmer DA. Angiogenesis in the placenta. Biol Reprod 2001;64:1033–1040. - PubMed
-
- Silha JV, Krsek M, Sucharda P, Murphy LJ. Angiogenic factors are elevated in overweight and obese individuals. Int J Obes Disord 2005;29:1308–1314. - PubMed
-
- Khurana R, Simons M, Martin JF, Zachary IC. Role of angiogenesis in cardiovascular disease: a critical appraisal. Circulation 2005;112:1813–1824. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

