The avian malaria parasite Plasmodium gallinaceum causes marked structural changes on the surface of its host erythrocyte
- PMID: 18442920
- PMCID: PMC2486836
- DOI: 10.1016/j.jsb.2008.03.005
The avian malaria parasite Plasmodium gallinaceum causes marked structural changes on the surface of its host erythrocyte
Abstract
Using a combination of atomic force, scanning and transmission electron microscopy, we found that avian erythrocytes infected with the avian malaria parasite Plasmodium gallinaceum develop approximately 60 nm wide and approximately 430 nm long furrow-like structures on the surface. Furrows begin to appear during the early trophozoite stage of the parasite's development. They remain constant in size and density during the course of parasite maturation and are uniformly distributed in random orientations over the erythrocyte surface. In addition, the density of furrows is directly proportional to the number of parasites contained within the erythrocyte. These findings suggest that parasite-induced intraerythrocytic processes are involved in modifying the surface of host erythrocytes. These processes may be analogous to those of the human malaria parasite P. falciparum, which induces knob-like protrusions that mediate the pathogenic adherence of parasitized erythrocytes to microvessels. Although P. gallinaceum-infected erythrocytes do not seem to adhere to microvessels in the host chicken, the furrows might be involved in the pathogenesis of P. gallinaceum infections by some other mechanism involving host-pathogen interactions.
Figures
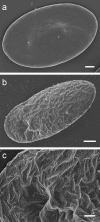


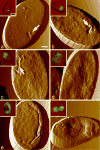
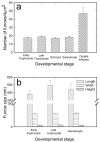

Similar articles
-
De novo assembly and transcriptome analysis of Plasmodium gallinaceum identifies the Rh5 interacting protein (ripr), and reveals a lack of EBL and RH gene family diversification.Malar J. 2015 Aug 5;14:296. doi: 10.1186/s12936-015-0814-0. Malar J. 2015. PMID: 26243218 Free PMC article.
-
Transcriptome sequencing and analysis of Plasmodium gallinaceum reveals polymorphisms and selection on the apical membrane antigen-1.Malar J. 2014 Sep 26;13:382. doi: 10.1186/1475-2875-13-382. Malar J. 2014. PMID: 25261185 Free PMC article.
-
A microfluidic platform to isolate avian erythrocytes infected with Plasmodium gallinaceum malaria parasites based on surface morphological changes.Biomed Microdevices. 2011 Dec;13(6):995-1004. doi: 10.1007/s10544-011-9569-8. Biomed Microdevices. 2011. PMID: 21769638
-
Membrane modifications in erythrocytes parasitized by Plasmodium falciparum.Mol Biochem Parasitol. 1996 Feb-Mar;76(1-2):1-10. doi: 10.1016/0166-6851(95)02575-8. Mol Biochem Parasitol. 1996. PMID: 8919990 Review.
-
Vesicle-mediated trafficking of parasite proteins to the host cell cytosol and erythrocyte surface membrane in Plasmodium falciparum infected erythrocytes.Int J Parasitol. 2001 Oct;31(12):1381-91. doi: 10.1016/s0020-7519(01)00256-9. Int J Parasitol. 2001. PMID: 11566305 Review.
Cited by
-
A member of the tryptophan-rich protein family is required for efficient sequestration of Plasmodium berghei schizonts.PLoS Pathog. 2022 Sep 20;18(9):e1010846. doi: 10.1371/journal.ppat.1010846. eCollection 2022 Sep. PLoS Pathog. 2022. PMID: 36126089 Free PMC article.
-
Is there a direct role for erythrocytes in the immune response?Vet Res. 2011 Jul 29;42(1):89. doi: 10.1186/1297-9716-42-89. Vet Res. 2011. PMID: 21801407 Free PMC article. Review.
-
Physicochemical Aspects of the Plasmodium chabaudi-Infected Erythrocyte.Biomed Res Int. 2015;2015:642729. doi: 10.1155/2015/642729. Epub 2015 Oct 18. Biomed Res Int. 2015. PMID: 26557685 Free PMC article.
-
Identification and expression of maebl, an erythrocyte-binding gene, in Plasmodium gallinaceum.Parasitol Res. 2013 Mar;112(3):945-54. doi: 10.1007/s00436-012-3211-4. Epub 2012 Dec 7. Parasitol Res. 2013. PMID: 23224610 Free PMC article.
-
Neuroimmunological blood brain barrier opening in experimental cerebral malaria.PLoS Pathog. 2012;8(10):e1002982. doi: 10.1371/journal.ppat.1002982. Epub 2012 Oct 25. PLoS Pathog. 2012. PMID: 23133375 Free PMC article.
References
-
- Aikawa M, Hsieh CL, Miller LH. Ultrastructural changes of the erythrocyte membrane in ovale-type malarial parasites. J Parasitol. 1977;63:152–154. - PubMed
-
- Aikawa M, Iseki M, Barnwell JW, Taylor D, Oo MM, Howard RJ. The pathology of human cerebral malaria. Am J Trop Med Hyg. 1990;43:30–37. - PubMed
-
- Aikawa M, Pongponratn E, Tegoshi T, Nakamura K, Nagatake T, Cochrane A, Ozaki LS. A study on the pathogenesis of human cerebral malaria and cerebral babesiosis. Mem Inst Oswaldo Cruz. 1992;87(Suppl 3):297–301. - PubMed
-
- Arie T, Fairhurst RM, Brittain NJ, Wellems TE, Dvorak JA. Hemoglobin C modulates the surface topography of Plasmodium falciparum-infected erythrocytes. J Struct Biol. 2005;150:163–169. - PubMed
-
- Aviado DM. Chemotherapy of Plasmodium berghei. Exp Parasitol. 1969;25:399–482. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

