An extracellular Cu2+ binding site in the voltage sensor of BK and Shaker potassium channels
- PMID: 18443360
- PMCID: PMC2346571
- DOI: 10.1085/jgp.200809980
An extracellular Cu2+ binding site in the voltage sensor of BK and Shaker potassium channels
Abstract
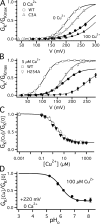
Copper is an essential trace element that may serve as a signaling molecule in the nervous system. Here we show that extracellular Cu2+ is a potent inhibitor of BK and Shaker K+ channels. At low micromolar concentrations, Cu2+ rapidly and reversibly reduces macrosocopic K+ conductance (G(K)) evoked from mSlo1 BK channels by membrane depolarization. GK is reduced in a dose-dependent manner with an IC50 and Hill coefficient of 2 microM and 1.0, respectively. Saturating 100 microM Cu2+ shifts the GK-V relation by +74 mV and reduces G(Kmax) by 27% without affecting single channel conductance. However, 100 microM Cu2+ fails to inhibit GK when applied during membrane depolarization, suggesting that Cu2+ interacts poorly with the activated channel. Of other transition metal ions tested, only Zn2+ and Cd2+ had significant effects at 100 microM with IC(50)s > 0.5 mM, suggesting the binding site is Cu2+ selective. Mutation of external Cys or His residues did not alter Cu2+ sensitivity. However, four putative Cu2+-coordinating residues were identified (D133, Q151, D153, and R207) in transmembrane segments S1, S2, and S4 of the mSlo1 voltage sensor, based on the ability of substitutions at these positions to alter Cu2+ and/or Cd2+ sensitivity. Consistent with the presence of acidic residues in the binding site, Cu2+ sensitivity was reduced at low extracellular pH. The three charged positions in S1, S2, and S4 are highly conserved among voltage-gated channels and could play a general role in metal sensitivity. We demonstrate that Shaker, like mSlo1, is much more sensitive to Cu2+ than Zn2+ and that sensitivity to these metals is altered by mutating the conserved positions in S1 or S4 or reducing pH. Our results suggest that the voltage sensor forms a state- and pH-dependent, metal-selective binding pocket that may be occupied by Cu2+ at physiologically relevant concentrations to inhibit activation of BK and other channels.
Figures
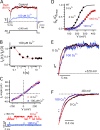
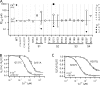
 ) and 200 μM (⋄), are normalized by GKmax in 0 Cu2+ and fit by Boltzmann functions. (B) Mean GK-V relations from panel A are normalized by GKmax at each [Cu2+] based on Boltzmann fits. (C) Cu2+ dose–response relations for ΔV0.5 = (V0.5 [Cu2+] − V0.5[0]) (•) and GK[Cu2+] at 110 mV (○), 150 mV (▵) and 190 mV (▿) were determined from fits in A. Solid lines are fits to a Hill equation. Thick dotted curve is a fit to Eq. 1 with most parameters set to previously determined values (zJ = 0.58 e, L0 = 10−6, zL = 0.3 e, KD(Ca2+) = 11 μM, C = 8, D = 25, E = 2.4) (Horrigan and Aldrich, 2002). VhC = 162 mV was adjusted to fit V0.5 of the 0 Cu2+ control, and Cu2+-dependent parameters (KDcu = 0.75 μM, Ecu = 0.124) were then varied to fit the V0.5-[Cu2+] relation. Thin dotted curve is a fit to Eq. 2 with KDcu = 2.1 μM, Ecu = 0.129, and all other parameters the same as for Eq. 1. (D) IC50 and (E) nH obtained by fitting GK-[Cu2+] relations in 3 μM Ca2+ (•) and 0 Ca2+ (□) are plotted at different voltages. Dashed lines are IC50 = 1.97 μM, nH = 1.01 from the ΔV0.5-[Cu2+] relation fit in panel C. (F) Apparent charge (Zapp) from Boltzmann GK-V fits in A are plotted versus [Cu2+] and fit by a Hill equation (IC50 = 0.72 ± 0.12 μM, nH = 1.1 ± 0.2). Curves A and B are the predictions of Eqs. 1 and 2, respectively, using parameters from C.
) and 200 μM (⋄), are normalized by GKmax in 0 Cu2+ and fit by Boltzmann functions. (B) Mean GK-V relations from panel A are normalized by GKmax at each [Cu2+] based on Boltzmann fits. (C) Cu2+ dose–response relations for ΔV0.5 = (V0.5 [Cu2+] − V0.5[0]) (•) and GK[Cu2+] at 110 mV (○), 150 mV (▵) and 190 mV (▿) were determined from fits in A. Solid lines are fits to a Hill equation. Thick dotted curve is a fit to Eq. 1 with most parameters set to previously determined values (zJ = 0.58 e, L0 = 10−6, zL = 0.3 e, KD(Ca2+) = 11 μM, C = 8, D = 25, E = 2.4) (Horrigan and Aldrich, 2002). VhC = 162 mV was adjusted to fit V0.5 of the 0 Cu2+ control, and Cu2+-dependent parameters (KDcu = 0.75 μM, Ecu = 0.124) were then varied to fit the V0.5-[Cu2+] relation. Thin dotted curve is a fit to Eq. 2 with KDcu = 2.1 μM, Ecu = 0.129, and all other parameters the same as for Eq. 1. (D) IC50 and (E) nH obtained by fitting GK-[Cu2+] relations in 3 μM Ca2+ (•) and 0 Ca2+ (□) are plotted at different voltages. Dashed lines are IC50 = 1.97 μM, nH = 1.01 from the ΔV0.5-[Cu2+] relation fit in panel C. (F) Apparent charge (Zapp) from Boltzmann GK-V fits in A are plotted versus [Cu2+] and fit by a Hill equation (IC50 = 0.72 ± 0.12 μM, nH = 1.1 ± 0.2). Curves A and B are the predictions of Eqs. 1 and 2, respectively, using parameters from C.

 ,
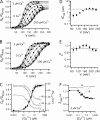
,  ; Ala: ○, •; Lys: □; Gln: ⋄; Asn: ⋄; Cys: ▿,▾; His: ▵, ▴). Dashed line indicates the IC50 of the WT for Cu2+. Vertical solid lines indicate the differences in IC50 between mutants at putative coordination sites. Dose–response relations (GK-[Cu2+]) for putative coordination sites 151 and 207 in 0 Ca2+ are plotted and fit by Hill equations in (B) Q151A (○, IC50 = 23.8 μM, nH =1.0), Q151C (<, IC50 = 0.96 μM, nH = 0.97) at +240 mV and (C) R207Q (⋄, IC50 = 85.8 μM, nH = 0.77) and R207C (<, IC50 = 0.52 μM, nH = 0.77) at +180 mV.
; Ala: ○, •; Lys: □; Gln: ⋄; Asn: ⋄; Cys: ▿,▾; His: ▵, ▴). Dashed line indicates the IC50 of the WT for Cu2+. Vertical solid lines indicate the differences in IC50 between mutants at putative coordination sites. Dose–response relations (GK-[Cu2+]) for putative coordination sites 151 and 207 in 0 Ca2+ are plotted and fit by Hill equations in (B) Q151A (○, IC50 = 23.8 μM, nH =1.0), Q151C (<, IC50 = 0.96 μM, nH = 0.97) at +240 mV and (C) R207Q (⋄, IC50 = 85.8 μM, nH = 0.77) and R207C (<, IC50 = 0.52 μM, nH = 0.77) at +180 mV.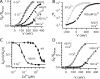
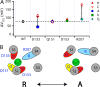
 ) and mutants at the four putative Cu2+-coordinating sites (Ala: •; Lys: ▪; Gln: ⋄; Cys: ▿; His: ▴). The dashed line indicates ΔV0.5MAX for the WT. The vertical solid lines indicate the range of ΔV0.5MAX for mutants at each position. (B) A speculative model of the Cu2+ binding site in the resting (R) and activated (A) state of the voltage sensor. In the resting state, Cu2+ is coordinated by D133(S1), Q151(S2), D153(S2), and R207(S4). The relative size and position of transmembrane segments correspond to those in the structure of a Kv1.2/Kv2.1 chimera (Long et al., 2007). During activation, the S2 residues interact with Cu2+ in a state-independent manner while interactions with D133 and R207 are weakened or disrupted, presumably by changes in the position or orientation of S1 and S4 relative to S2. In this way, mutation of any of these residues alter IC50, but only mutation of D133 or R207 alter efficacy.
) and mutants at the four putative Cu2+-coordinating sites (Ala: •; Lys: ▪; Gln: ⋄; Cys: ▿; His: ▴). The dashed line indicates ΔV0.5MAX for the WT. The vertical solid lines indicate the range of ΔV0.5MAX for mutants at each position. (B) A speculative model of the Cu2+ binding site in the resting (R) and activated (A) state of the voltage sensor. In the resting state, Cu2+ is coordinated by D133(S1), Q151(S2), D153(S2), and R207(S4). The relative size and position of transmembrane segments correspond to those in the structure of a Kv1.2/Kv2.1 chimera (Long et al., 2007). During activation, the S2 residues interact with Cu2+ in a state-independent manner while interactions with D133 and R207 are weakened or disrupted, presumably by changes in the position or orientation of S1 and S4 relative to S2. In this way, mutation of any of these residues alter IC50, but only mutation of D133 or R207 alter efficacy.
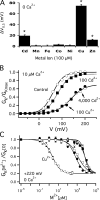
 )). (E) IK at 0 mV from E247A channels in 0, 1, and 5 μM Cu2+. (F) IK at +30 mV from E247C in 0, 0.1, 1, and 5 μM Cu2+. (G and H) IK at +10 mV from E247A (G) or E247C (H) in 0, 100, and 1,000 μM Zn2+. (I and J) IK from R365A at 0 mV (I) or R365C at −40 mV(J) in 0, 1, and 5 μM Cu2+. All Shaker experiments were performed from a holding potential of −90 mV. Most data were obtained in standard Shaker internal and external solutions (see Materials and methods) with the exception of R365 mutant data in I and J, which were obtained in a low chloride external solution with all but 10 mM Cl− replaced by MeO3−, as in standard mSlo1 external solution.
)). (E) IK at 0 mV from E247A channels in 0, 1, and 5 μM Cu2+. (F) IK at +30 mV from E247C in 0, 0.1, 1, and 5 μM Cu2+. (G and H) IK at +10 mV from E247A (G) or E247C (H) in 0, 100, and 1,000 μM Zn2+. (I and J) IK from R365A at 0 mV (I) or R365C at −40 mV(J) in 0, 1, and 5 μM Cu2+. All Shaker experiments were performed from a holding potential of −90 mV. Most data were obtained in standard Shaker internal and external solutions (see Materials and methods) with the exception of R365 mutant data in I and J, which were obtained in a low chloride external solution with all but 10 mM Cl− replaced by MeO3−, as in standard mSlo1 external solution.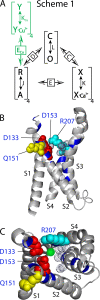
Similar articles
-
Role of charged residues in the S1-S4 voltage sensor of BK channels.J Gen Physiol. 2006 Mar;127(3):309-28. doi: 10.1085/jgp.200509421. J Gen Physiol. 2006. PMID: 16505150 Free PMC article.
-
Role of hydrophobic and ionic forces in the movement of S4 of the Shaker potassium channel.Mol Membr Biol. 2012 Dec;29(8):321-32. doi: 10.3109/09687688.2012.710343. Epub 2012 Aug 13. Mol Membr Biol. 2012. PMID: 22881396
-
Effect of cysteine substitutions on the topology of the S4 segment of the Shaker potassium channel: implications for molecular models of gating.J Physiol. 1999 Dec 1;521 Pt 2(Pt 2):315-26. doi: 10.1111/j.1469-7793.1999.00315.x. J Physiol. 1999. PMID: 10581304 Free PMC article.
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Large conductance Ca2+-activated K+ (BK) channel: activation by Ca2+ and voltage.Biol Res. 2006;39(3):385-401. doi: 10.4067/s0716-97602006000300003. Epub 2006 Nov 7. Biol Res. 2006. PMID: 17106573 Review.
Cited by
-
Threading the biophysics of mammalian Slo1 channels onto structures of an invertebrate Slo1 channel.J Gen Physiol. 2017 Nov 6;149(11):985-1007. doi: 10.1085/jgp.201711845. Epub 2017 Oct 12. J Gen Physiol. 2017. PMID: 29025867 Free PMC article. Review.
-
Dietary cholesterol modulates the excitability of rabbit hippocampal CA1 pyramidal neurons.Neurosci Lett. 2010 Aug 2;479(3):327-31. doi: 10.1016/j.neulet.2010.05.090. Neurosci Lett. 2010. PMID: 20639007 Free PMC article.
-
Metal Bridge in S4 Segment Supports Helix Transition in Shaker Channel.Biophys J. 2020 Feb 25;118(4):922-933. doi: 10.1016/j.bpj.2019.08.035. Epub 2019 Sep 5. Biophys J. 2020. PMID: 31635788 Free PMC article.
-
Incorporating physics to overcome data scarcity in predictive modeling of protein function: A case study of BK channels.PLoS Comput Biol. 2023 Sep 15;19(9):e1011460. doi: 10.1371/journal.pcbi.1011460. eCollection 2023 Sep. PLoS Comput Biol. 2023. PMID: 37713443 Free PMC article.
-
Inactivation of TRPM2 channels by extracellular divalent copper.PLoS One. 2014 Nov 11;9(11):e112071. doi: 10.1371/journal.pone.0112071. eCollection 2014. PLoS One. 2014. PMID: 25386648 Free PMC article.
References
-
- Assaf, S.Y., and S.H. Chung. 1984. Release of endogenous Zn2+ from brain tissue during activity. Nature. 308:734–736. - PubMed
-
- Bewley, M.C., P.D. Jeffrey, M.L. Patchett, Z.F. Kanyo, and E.N. Baker. 1999. Crystal structures of Bacillus caldovelox arginase in complex with substrate and inhibitors reveal new insights into activation, inhibition and catalysis in the arginase superfamily. Structure. 7:435–448. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

